When an amine is protonated, the resulting ammonium ion is not electrophilic: However, when an imine is
Question:
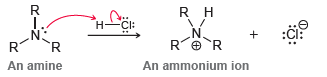
However, when an imine is protonated, the resulting iminium ion is highly electrophilic:
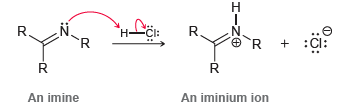
Explain this difference in reactivity between an ammonium ion and an iminium ion.
Transcribed Image Text:
R H CI: Н- 'R An amine An ammonium ion N. CI: R. :Cl: An imine An iminium ion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
The nitrogen atom of an ammonium ion is positively charged but that does not render it ...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When aniline is treated with a mixture of nitric acid and sulfuric acid, the expected nitration product (para-nitroaniline) is obtained in poor yield. Instead, the major product from nitration is...
-
The benzyl ammonium ion (C6H5CH2NH3) has pKa 9.33, and the propyl ammonium ion has pKa = 10.71. Which is the stronger base, benzyl amine or propylamine? What are the pKbS of benzyl amine and...
-
The ammonium ion, NH4+, has a tetrahedral geometry analogous to that of methane. Explain this structure in terms of atomic and molecular orbitals.
-
Solve the inequality. Write the solution in interval notation. |-3x + 1 5
-
Evaluate business risk and any debt/equity decisions recently made for Spirit Airlines Inc. Identify at least three business risks of Spirit Airlines Inc. may face in the next year. 1. Do the...
-
Two newly hired employees at Gordian Yacht, a luxury boat manufacturer, are beginning their probationary period on the job. Their supervisor assesses a .6 proba-bility that employee A will make it...
-
A unidirectional \(45^{\circ}\) off-axis E-glass/epoxy composite lamina is supported on frictionless rollers between rigid walls as shown in Figure 5.18. The lamina is fixed against displacements in...
-
As the executive of a bank or thrift institution you are faced with an intense seasonal demand for loans. Assuming that your loanable funds are inadequate to take care of the demand, how might your...
-
The Tom Corp wants to know its cost of capital. Its current capital structure calls for 4 5 % debt, 1 5 % preferred stock and 4 0 % common equity. Initially, common stock will be in the form of...
-
A 46 year old male was found unconscious in Nottingham City centre at 1pm and when ambulance staff tried to wake him he was unresponsive Upon admission to accident and emergency at QMC, the patient...
-
Consider the following reaction. Predict whether an increase in temperature will favor reactants or products. Justify your prediction. +
-
Draw the curved arrows that accomplish the following transformation: CI CI H. e Me Me NEN Me
-
Every large organization has large files or databases containing data used in operating the business. How does a data warehouse differ from these operational files or databases? Why are these...
-
What are the two primary tests that good cost allocation processes pass?
-
Describe the ways a salesperson is able to add value through customer follow-up.
-
How does the statement differ between not-for-profit and forprofit entities?
-
List and briefly describe the golden rules for success in formal negotiations.
-
What are the reasons for a follow-up with customers, and why do they add value?
-
Suppose that indifference curves are described by straight lines with a slope ofb. Given arbitrary prices and money income p 1 , p 2 , and m, what will the consumers optimal choices look like?
-
What is a lobbyist in US? How did this term emerge?
-
(a) Outline a step-by-step mechanism for the phenylation of acetoacetic ester by bromobenzene and two molar equivalents of sodium amide. (Why are two molar equivalents of NaNH2 necessary?) (b) What...
-
Rank the following in order of increasing acidity. NO2 CF3 CF3
-
Without consulting tables, select the stronger acid from each of the following pairs: (a) 4-Methylphenol and 4-fluorophenol (b) 4-Methylphenol and 4-nitrophenol (c) 4-Nitrophenol and 3-nitrophenol...
-
What is the balance in Work in Process Inventory at the end of each month? Work in Process Inventory April 30 $ May 31 A 10,140 15,950 June 30 $ 10,450
-
Sako Company s Audio Division produces a speaker used by manufacturers of various audio products. Sales and cost data on the speaker follow: Selling price per unit on the intermediate market $ 6 0...
-
Vulcan Company's contribution format income statement for June is as follows: Vulcan Company Income Statement For the Month Ended June 30 Sales Variable expenses Contribution margin Fixed expenses...

Study smarter with the SolutionInn App


