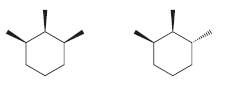
Consider the following two compounds. These compounds are stereoisomers of 1,2,3-trimethylcyclohexane. One of these compounds has three
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
The first compound has three chirality centers This is apparent if we assi...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite rapidly, while the other forms an epoxide very slowly. Identify which compound reacts more...
-
Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1 H NMR spectroscopy? c) 13 C NMR spectroscopy?
-
Consider the following two compounds: CH3CH2CH2CH2CH2OH 1-pentanol CH3CH2CH2CH2CH2CH3 hexane a. What are the different types of intermolecular forces that exist in each compound? b. One of these...
-
. What impacts of influences adult human behavior? Perspective Support: You must critically evaluate the issue and address the question: " What influences or impacts personality development?" by...
-
What role do finance companies play in leveraged buyouts?
-
Write expressions for K and Kp for the following reactions. a. 2NH3(g) + CO2(g) N2CH4O(s) + H2O(g) b. 2NBr3(s) N2(g) + 3Br2(g) c. 2KClO3(s) 2KCl(s) + 3O2(g) d. CuO(s) + H2(g) Cu(l) + H2O(g)
-
After installation, the lifetime of a ball bearing is modeled as an exponential distribution with failure rate \(\alpha=0.001\) failures per day. (a) What is the probability that the ball bearing...
-
1. Who are the stakeholders in this case, and what are their stakes? 2. What social responsibility does the firm have for the environment? How would you assess the firms CSR using the four-part CSR...
-
What would be good thorough answers to the below questions? The role of the financial manager is crucial inevery type of organization. Locate a job posting for a financial manager position from any...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Cyclopropane is a compound in which the carbon atoms form a three-membered ring: Each of the carbon atoms in cyclopropane is sp 3 hybridized. Cyclopropane is more reactive than other cyclic compounds...
-
When butyl bromide is treated with sodium iodide in ethanol, the concentration of iodide quickly decreases but then slowly returns to its original concentration. Identify the major product of the...
-
Write a MATLAB program that can be used to find the range of sampling time, T, for stability. The program will be used for systems of the type represented in Figure P13.5 and should meet the...
-
Suppose that the quality control engineer in question 35 does not know what the standard deviation is and therefore uses the sample standard deviation. Does your answer to question 35 change? Why or...
-
Suppose that the quality control engineer in question 35 does not know what the standard deviation is and that this time, he selects a random sample of only 25 light bulbs. Does your answer to...
-
Use the information given in question 38 to test the hypothesis that the population mean accounts receivable is greater than $200 at the .05 level of significance. Question 38 An auditor is...
-
An investment advisor is interested in determining whether a retirement community represents a potential clientele base. Of the 2,000 residents, he randomly samples 100 individuals and finds their...
-
Write pros and cons of Heap-Sort, Merge-Sort and Quick-Sort.
-
Which of the following would not generally appear on the M-1 reconciliation? a. Federal income tax expense per books. b. Tax-exempt interest income. c. Excess tax over book depreciation. d. Dividends...
-
From the choice of simple moving average, exponential smoothing, and linear regression analysis, which forecasting technique would you consider the most accurate? Why? please write it in word...
-
What products would you expect from oxidation of the following compounds with CrO3 in aqueous acid with pyridinium chlorochromate? (a) 1-Hexanol (b) 2-Hexanol (c) Hexanol
-
TMS ethers can be removed by treatment with fluoride ion as well as be acid-catalyzed hydrolysis. Propose a mechanism for the reaction of cyclohexyl TMS ether with LiF. Fluorotrimethyisilane is a...
-
Show the mechanism of the reaction of p-methaylphenol with 2-methylpropene and H3PO4 catalyst to yield the food additive BHT.
-
A consultant advises that a fund pays out $100,000 at the end of 5 years. Provided $12,000 is invested in the fund at the start of each year, beginning immediately and continuing up to and including...
-
Durco Automotive needs a $2 million balance in its contingency fund 3 years from now. The chief financial officer (CFO) wants to know how much to deposit now into Durco's high-yield investment...
-
3. Create a chart for gross margin less R&D and capital costs calculations. a. Calculate gross margin less R&D and capital costs for options A and B. b. Graph total gross margin less R&D and capital...

Study smarter with the SolutionInn App


