Consider the structures of the following alkyl chlorides: a. Which compound would you expect to undergo an
Question:
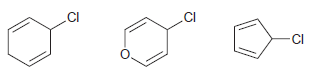
a. Which compound would you expect to undergo an SN1 process most readily? Justify your choice.
b Which compound would you expect to undergo an SN1 process least readily? Justify your choice.
Transcribed Image Text:
.CI .CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
a Loss of the leaving gr...View the full answer

Answered By

Subash Murugaih
I am leading expert in this web site couple of years and My clients are much happy with my works and services.
4.60+
309+ Reviews
539+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the structures of the following two d-aldotetroses: Each of these compounds exists as a furanose ring, which is formed when the OH at C 4 attacks the aldehyde moiety. Draw each of the...
-
Consider the structures of the four d-aldopentoses (See the following figure). (a) Which d-aldopentose produces the same aldaric acid as d-lyxose? (b) Which d-aldopentoses yield optically inactive...
-
Consider the structures of the constitutional isomers, Compound A and Compound B (below). When treated with aqueous acid, Compound A undergoes isomerization to give a cis stereoisomer. In contrast,...
-
For each of the following tests, identify two different samples of people who would have the expertise to serve as subject matter experts (SMEs) for providing judgments regarding the content validity...
-
What advice would you offer an entrepreneur interested in launching a global business effort? (LO 2: Describe the nine principal strategies small businesses can use to go global; AACSB: Reflective...
-
One strategy for producing a protein in the milk of a cow is to place the coding sequence of the gene of interest next to a __________ and then inject the gene into a _________. a. lac operon...
-
When should the data preparation process start and why?
-
Newland Company reported retained earnings at December 31, 2016, of $310,000. Newland had 200,000 shares of common stock outstanding at the beginning of 2017. The following transactions occurred...
-
Riverbend Incorporated received a $ 2 2 2 , 5 0 0 dividend from stock it held in Hobble Corporation. Riverbend's taxable income is $ 2 , 8 0 0 , 0 0 0 before deducting the dividends - received...
-
On December 1, Jasmin Ernst organized Ernst Consulting. On December 3, the owner contributed $84,000 in assets in exchange for its common stock to launch the business. On December 31, the companys...
-
Could cash-to-cash cycle time be negative? How? Would that be good?
-
What would be some good inventory management performance measures for a fast-food company? A bicycle repair shop? A big-box retailer?
-
The United States and China are the world's two largest economies, and their trade and investment linkages are expanding. Although the two countries have sought to cooperate on many topics regarding...
-
A club consists of 16 men and 19 women. In how many ways can they choose a president, vice president, treasurer, and secretary, along with an advisory committee of six people (who are not officers)?
-
In Problems 12-15, match each formula in Column A with the type of financial problem in Column B. Column A 15. m = 1 P() - (1 + 2) -nt Column B Sinking fund
-
A speed control system of an engine is shown in Fig. P1.2. Determine: (a) Sensitivity of closed loop system to changes in engine gain \(k_{1}\) and tachometer feedback gain \(k_{2}\). (b) Steady...
-
Use a calculator to evaluate an ordinary annuity formula \[A=m\left[\frac{\left(1+\frac{r}{n}ight)^{n t}-1}{\frac{r}{n}}ight]\] for \(m, r\), and \(t\) (respectively) given in Problems 7-22. Assume...
-
Find the present value of the ordinary annuities in Problems 21-32. Amount of Deposit m 32. $100 Frequency n quarterly Rate r 3% Time t 20 yr
-
Apply the methods of this and the previous section to graph each function. Be sure to label key points and show at least two periods. y = 1/2 cot(2x - )
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.) C,H OH/H,O CH CH2 sodium azide
-
Suggest a Williamron other cynthosis, if one is possible, for each of the following compounds. If no Williamson ether synthesis is possible, explain why. (CH3)2CH---S---CH3
-
Show how the stereochemistry of the products will differ (if at all) when the following enantiomerically pure epoxide is hydrolyzed under acidic and basic conditions. D,C
-
The following information is available for ADT Company, which produces special-order security produc order costing system. Overhead is applied using a predetermined overhead rate of 55% of direct...
-
Stahelin Valves produces a single component, a valve. The valve sells for $43 per unit. Fixed costs are $1,605,000 annually. Production and sales of 407,000 units annually results in profit before...
-
Calaveras Tire exchanged equipment for two pickup trucks. The book value and fair value of the equipment given up were $29,000 (original cost of $78,500 less accumulated depreciation of $49,500) and...

Study smarter with the SolutionInn App


