Count the total number of s bonds and p bonds in the compound below: -3-N
Question:
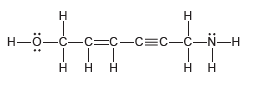
Transcribed Image Text:
н Н Н—ӧ—с—с-с—С3с—с-N—Н ннн н Н нн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Sixteen ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Show that the total number of atomic states (including different spin states) in a shell of principal quantum number n is 2n2. (b) Which shell has 50 states?
-
Figure gives a versus the sine of the angle in a single-slit diffraction experiment using light of wavelength 610 nm. The vertical axis scale is set by as = 12 rad. What are? (a) The slit width, (b)...
-
Figure gives the parameter B of Eq. 36-20 versus the sine of the angle in a two-slit interference experiment using light of wavelength 435nm. The vertical axis scale is set by s = 80.0 rad what are?...
-
Simplify the given expressions. Express all answers with positive exponents. 25/7/7/4 2/3 a
-
Write a report which should cover the Financial Statement Analysis for AT&T Inc. using Financial Ratios
-
A typical raindrop is much more massive than a mosquito and \(\mathrm{BO}\) falling much faster than a mosquito flies. How does a mosquito survive the impact? Recent research has found that the...
-
Problem 3.8 asked you to fit two different models to the chemical process data in Table B.5. Perform appropriate residual analyses for both models. Discuss the results of these analyses. Calculate...
-
Pcost Company purchased 85% of the common stock of Scost Company on April 1, Year 1. The fair value of the consideration transferred consisted of a cash payment of $545,000 and contingent...
-
1. Compute the activity rate for each activity using activity-based costing. 2. Compute overhead cost per unit for each of the two products using activity-based costing. 3. Determine product cost per...
-
The JMR Company is a family business that currently uses no debt in its capital structure. The owner-managers agreed on a plan to issue a large amount of debt to expand the company's operations....
-
Identify the expected hybridization state and geometry for the central atom in each of the following compounds: a. b. c. d. e. H. I-z: I-
-
Calculate the standard enthalpy of formation of FeS 2 (s) at 600. °C from the following data at 298.15 K. Assume that the heat capacities are independent of temperature. You are also given that...
-
The City of Ronchester has a defined benefit pension plan for its firefighters. How is the amount of pension expense determined that should be recognized in the current year?
-
What is the value of Michaels capital at the end of 2022? Michael, an Internet service provider, bought an existing business worth $400,000 on December 31, 2021. During 2022, his business grew and he...
-
If the desired reserve ratio and the currency drain ratio increase, how does the money multiplier change?
-
Explain the flows of funds that finance business investment.
-
Is the United States a net borrower or a net lender? Is it a debtor or a creditor nation?
-
Explain why doctors are paid more than nurses. According to a 2012 ABS Survey of Employee Earnings and Hours, full-time doctors in Australia earned a weekly average of AUD2,862.30 and full-time...
-
In problem, f() = sin and g() = cos . Find the exact value of each function below if = 60. Do not use a calculator. g()
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
Provide a mechanism that explains formation of the following products. CI HCI (concdCI+
-
Provide a mechanism that explains formation of the following products. OMe MeOH +CI OMet
-
Treating either 1-chloro-3-methyl-2-butene or 3-chloro-3-methyl-1-butene with Ag2O in water gives (in addition to AgCl) the following mixture of alcohol products. (a) Write a mechanism that accounts...
-
How might technology change in the future? How will technology make our lives better in the next ten years? What kinds of technology are you most excited about? What do you think will be the next...
-
How do the local electronic and printing advertisements apply in the real ground? What are the most accepted brand names? What are the good qualities and demerits of the brand?
-
Gary and Linda assume they will need $70,000 at the beginning of each year in today's dollars when they retire in 6 years. The couple is assuming an annual inflation rate of 4% and a 7.5% after-tax...

Study smarter with the SolutionInn App


