DEET is the active ingredient in many insect repellants, such as OFF TM . Starting with meta-bromotoluene
Question:
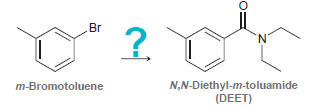
Transcribed Image Text:
.Br m-Bromotoluene N,N-Diethyl-m-toluamide (DEET)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Br 1 Mg ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
N, N-Diethyl-m-toluamide (DEFT) is the active ingredient in many insect- repellent preparations. Flow might you synthesize this substance fromm-bromotoluene? . CH2CH3 N,N-Diethyl-m-toluamide CH-CH
-
Acetylsalicylic acid is the active ingredient in aspirin. It took 35.17 mL of 0.5065 M sodium hydroxide to react completely with 3.210 g of acetylsalicylic acid. Acetyl-salicylic acid has one acidic...
-
Naproxen, a nonsteroidal anti-inflammatory drug, is the active ingredient in Aleve. Naproxen has a specific rotation + 66o of in chloroform. One commercial preparation results in a mixture that is...
-
In Figure 3-4, the current position of the demand curve is D 1 , and the price of a wireless earbud, which is a normal good, is $3. If there is an increase in consumer incomes, will the demand curve...
-
How strong are the competitive forces confronting Tiffany & Co. and other retail jewelers? Which one of the five competitive forces is the strongest? Do a five-forces analysis to support your answer.
-
Several data items that might be included on a payroll form are listed below on the left. The standard payroll forms discussed in the chapter are listed below on the right. For each data item,...
-
Construct a frequency histogram for the data set using seven classes. Use the data set shown in the table at the left, which represents the actual liquid volumes (in ounces) in 25 twelve-ounce cans....
-
The following transactions and adjusting entries were completed by Legacy Furniture Co. during a three-year period. All are related to the use of delivery equipment. The double-declining-balance...
-
Write a complete Python program that computes some measurements related to a swimming pool. Our pool does not have a shallow end, the depth is the same along the length of the pool. From a diving...
-
What is the solution of the recursive equation T (n) = 0.015625T +n?? (1/4) a) O(n') b) O(n' lg n) c) O(n* Ign) d) O(7*)
-
How did the Toyota Production System get started?
-
What is poka -yoke, who developed the concept, and what do you think it has to do with lean?
-
For the first-order reaction t 1/2 = 22.5 h at 20 C and 1.5 h at 40 C. (a) Calculate the activation energy of this reaction. (b) If the Arrhenius constant A = 2.05 x 10 13 s -1 , determine the value...
-
Identify one or more circumstances when a company might wish to delay introducing its product.
-
Name some industries in which a parallel process would not be possible or effective.
-
How can managers ensure that a team reaps the advantages of diversity while not being thwarted by some of the challenges team diversity raises?
-
A mixture that contains 40% by mole n-heptane in n-decane is to be separated in a series of flush separators until a stream is obtained that contains at least 95% n-heptane. Determine the number of...
-
Around $500 billion in U.S. Treasuries are traded each day and many countries hold U.S. Treasuries, as they are considered a very safe investment. For example, as of October 2018, Japan held $1,018.5...
-
Use synthetic division to decide whether the given number k is a zero of the polynomial function. If it is not, give the value of (k). (x) = x 3 + 3x 2 - x + 1; k = 1 + i
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
Show the 2p orbital's, and indicate the orbital overlap symbolized by the resonance structures for the carbocation in Eq. 15.32 on p. 711. Eq. 15.32 more important because each atom has a complete...
-
Show the 2p orbital's, and indicate the orbital overlap symbolized by the resonance structures for the carbocation in Eq. 15.32 on p. 711. Eq. 15.32 more important because each atom has a complete...
-
Using resonance arguments, state which ion or radical within each set is more stable. Explain. CHj HC-C CH2 or HC CH CH CH2
-
Develop a 3-year financial projection for your proposed cancer tele-genetics program . State any and all assumptions you make in creating your estimates. Please consider the following in creating...
-
Lets assume that we have project L, which has a 40% chance of turning out good. The company Raises $7,000, 50% from bondholders demanding a 6% interest rate and 50% from stockholders, which becomes...
-
Please assist in the filling out the above and below table. I have provided all information that has been provided to me. Company Shares Outstanding Latest Share Price MVE Total Debt W d W e a b c =...

Study smarter with the SolutionInn App


