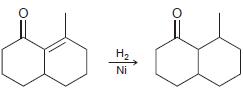
Describe how IR spectroscopy might be used to monitor the progress of each of the following reactions.
Question:
Describe how IR spectroscopy might be used to monitor the progress of each of the following reactions.
a.
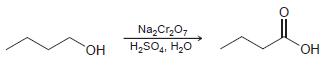
b.
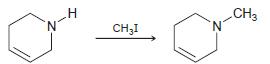
c.
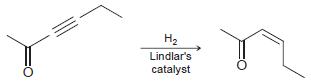
d.
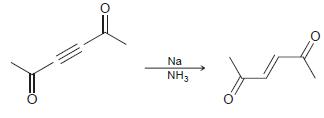
e.
Transcribed Image Text:
Na, Cr,0, H,SO,, H,0 HO, HO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a The starting material is an alcohol and is expected to produce a typical signal for an OH stretch ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify how IR spectroscopy might be used to monitor the progress of each of the following reactions. a. b. c. d. e. H2 Pt [0]
-
How can a multi-factor risk model be used to monitor and control portfolio risk?
-
Describe some ways in which technology might be used to ensure that computer users signing contracts with e-signatures are in fact the persons they are representing themselves to be.
-
Each team member is to become an expert on one depreciation method to facilitate teammates' understanding of that method. Follow these procedures: a. Each team member is to select an area of...
-
Under conditions assumed by the Classical economists, can the Fed control nominal interest rates? How about real interest rates?
-
Based on their bad-news communications, would you like to do business with these organizations?
-
For each of the following, calculate the standard error of the difference \(\left(s \mathrm{X}^{-} 1-\mathrm{X}^{-} ight.\) 2 ) and the \(t\)-test for independent means. a. N = 6, N = 6, X 1 18.50, $...
-
Here is a CPM network with activity times in weeks: a. Determine the critical path. b. How many weeks will the project take to complete? c. Suppose F could be shortened by two weeks and B by one...
-
Corporate) taxes play an important role in making investment decisions. Taxes influence the incremental cash flows needed to assess the feasibility of an investment. Explain why and how taxes are...
-
Shafer Company has gathered data on its overhead activities and associated costs for the past 10 months. Theodore, a member of the controllers department, has convinced management that overhead costs...
-
A stock market exists a. Only to service the sale of new issues, called IPOs. b. To provide liquidity to all stocks, including recent IPOs. c. Help policy makers predict the future. d. To make the...
-
What two numbers balance? a. The current account and exports. b. The capital account and the current account. c. Exports and imports. d. Short term investment income and short term investment...
-
Characterize ribosomes in general as to size, location, function, and macromolecular composition.
-
The instructions on the chlorinator for a fish tank say to put in five drops per gallon. Jeff has a fish tank that holds 2.27 ft. of water. How many drops to Jeff use
-
12) Compared to a methane molecule (CH4, FW = 16), the average speed of a sulfur dioxide molecule (SO2, FW = 64) is a) 4 times faster c) 2 times slower e) equally fast b) 2 times faster d) 4 times...
-
A brick wall 30 ft in length contains 2060 bricks. At the same rate, how many bricks would it take to build a wall 54 ft in length
-
line r has an equation of y + 1 0 = - 2 ( x - 6 ) . line s is perpendicular to line r and passes theough ( 1 , 2 ) . whqt is the equation of line s ?
-
If the population is 37,691,912 people, and the area is 158,633 mi2, what is the population density
-
In a random sample of 120 families that use day care, how many pay more than $7250 annually for day care per child? Assume the annual day care cost per child is normally distributed with a mean of...
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
How would you prepare the following diols? (b) (a)
-
Predict the major product of the following reactions: (a) (b) 18 CH-CH CH-CH NaOH H2C-o "H H2C- 18 CH (c) MgBr 1. CH-CH . 2. * "CH
-
15-Crown-5 and 12-crown-4 ethers complex Na + and Li +, respectively. Make models of these crown ethers, and compare the sized of the cavities.
-
Determine your monthly payment if you buy a $300,000 house at 6% with 15% down and pay it off over 30 years.
-
Revenue Recognition at a Point in Time versus Revenue Recognition Over Time. The market company won a contract to build a shopping center at a price of $300 million. The following schedule details...
-
Calculate the flat monthly instalment for the car loan amount of $450,000 over two years at 5% compounded semiannualy. You can use either geometric progression or an optimizer (Goal seek or Solver)...

Study smarter with the SolutionInn App


