Determine the hybridization state of each carbon atom in the following compounds: a. b. , C=c=c=C
Question:
a.
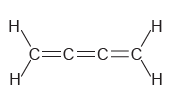
b.
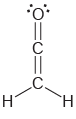
Transcribed Image Text:
Н, н C=c=c=C Н н || Н `H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
a b ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the hybridization state of each carbon in the following Compounds: (b) Formaldehyde (H2C=O) (c) Ketene (H2 C=C=O) (d) Propane (CH3CH=CH2) (e) Acetone [(CH3)2C=O] (f) Acrylonitrile (CH2=CHC CPN)
-
Give the hybridization state of each carbon in the following compounds: (b) Formaldehyde (H2C==O) (c) Ketene (H2C==C==O) (d) Propene (CH3CH==CH2) (e) Acetone [(CH3)2C==O] (f) Acrylonitrile (CH2==CHCN)
-
Below are the structures of two common over-the-counter pain relievers. Determine the hybridization state of each carbon atom in these compounds: a. b. TH. H H H. .C. TH. .C: .. `H Acetylsalicyclic...
-
In seawater, the pressure p is related to the depth d according to 33p - 18d = 495 where d is in feet and p is in pounds per square inch. (a) Solve this equation for p in terms of d. (b) The Titanic...
-
"You know, we recently had a soft drink product (an exotic berry seltzer line) go through one of those simulated test markets, and it was a disaster. The new products people forgot completely about...
-
Repeat Problem 9 assuming that floor support and vibration dampening must be added for the gang punch. These one-time first costs are estimated to be \(\$ 35,000\). Data From Problem 9 Bailey, Inc.,...
-
Consider the regression model in Problem 3.43. The value of the adjusted $R^{2}$ is Data From Problem 3.43 You have fit a linear regression model with three predictors to a sample of 50 observations....
-
The SEC Form 10-K of Google is reproduced online at www.wiley.com/college/pratt. REQUIRED: Review the 10-K, and answer the following questions: a. What are the major sources of cash for the company...
-
If the indirect method is used to calculate the intrinsic value of a company's equity. How to compare the intrinsic value per share and the value per share?
-
Assume you performed sampling for an accounts receivable population with a recorded population amount of $2,000,000. Tolerable misstatement is set at $100,000 for the test, and there are no...
-
Identify the reagents you would use to accomplish the following transformation. CI
-
At year-end 2009, total assets for Shome Inc. were $1.2 million and accounts payable were $375,000. Sales, which in 2009 were $2.5 million, are expected to increase by 25 percent in 2010. Total...
-
Tire and automobile manufacturers and consumer safety experts all recommend that drivers maintain proper tire pressure in their cars. Consequently, many gas stations now provide air pumps and air...
-
Differentiate between accelerator principle of investment and investment multiplier. Explain the determinants of induced investment in an economy. Given C=a+bY, explain "a" and "b" and then sketch...
-
What can you do to increase the performance of an embedded system?
-
. How a flash memory can be Read, Erased, or Programmed? Explain the steps involved.
-
List any four applications of KNN. Explain with Example
-
Explain Demings quality principles? with Example
-
Determine the mean, median, mode, and midrange of the set of data. Where appropriate, round your answer to the nearest tenth. 1, 1, 1, 1, 4, 4, 4, 4, 6, 8, 10, 12, 15, 21
-
Should U.S. antidumping laws be stated in terms of average total costs or average variable costs?
-
Draw an energy diagram for a reaction with Keq = 1. What is the value of G in this reaction?
-
The addition of water to ethylene to yield ethanol has the following thermodynamic parameters: (a) Is the reaction exothermic or endothermic? (b) Is the reaction favorable (spontaneous) or...
-
When a mixture of methane and chlorine is irradiated, reaction commences immediately. When irradiation is stopped, the reaction gradually slows down but does not stop immediately. Explain.
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


