The addition of water to ethylene to yield ethanol has the following thermodynamic parameters: (a) Is the
Question:
The addition of water to ethylene to yield ethanol has the following thermodynamic parameters:
(a) Is the reaction exothermic or endothermic?
(b) Is the reaction favorable (spontaneous) or unfavorable (non-spontaneous) at room temperature (298K)?
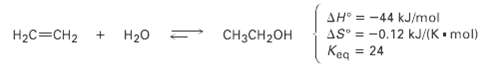
Transcribed Image Text:
AH° = -44 kJ/mol AS° = -0.12 kJ/(K• mol) = 24 %3! CHзCH20H H2C=CH2 + Н2о Квg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a The reaction is exothermic because t...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or the Si face of the...
-
Consider the following reaction: (a) Is this reaction exothermic or endothermic? (b) Calculate the amount of heat transferred when 3.55 g of Mg(s) reacts at constant pressure. (c) How many grams of...
-
Write an equation for the acid-catalyzed addition of water to a. 3-hexene b. 2-butene c. 4-methylcyclopentene
-
For the increasing marginal-extraction-cost model of the allocation of a depletable resource, how would the ultimate cumulative amount taken out of the ground be affected by (a) An increase in the...
-
Tim and Allison are married and have two children, ages 2 and 6. Allison is a "nonworking" spouse who devotes all of her time to household activities. Estimate how much life insurance Tim and Allison...
-
Consider the rest of the world with free trade (production at 5, and consumption at C,), as shown in the graph on the right in Figure 4.4A. The international relative price of cloth now changes to...
-
A carbon/epoxy single fiber test specimen is subjected to a uniaxial tensile stress that is increased until the fiber breaks up into pieces having a length of \(0.625 \mathrm{~mm}\). If the fiber has...
-
For its three investment centers, Paige Company accumulates the following data: Compute the return on investment (ROI) for eachcenter. III II Sales Controllable margin Average operating assets $...
-
What is community psychology (CP)?
-
On March 1, 2018, Maxus Energy acquired a gold mine for $29,000,000. At the time of acquisition, the Proven and Probable gold reserves of the mine were estimated to be 470,000 ounces. During the...
-
Draw an energy diagram for a reaction with Keq = 1. What is the value of G in this reaction?
-
When a mixture of methane and chlorine is irradiated, reaction commences immediately. When irradiation is stopped, the reaction gradually slows down but does not stop immediately. Explain.
-
Laura?s Fresh Cooking Inc. began operations on January 1, 2019. Laura?s prepares gourmet dinners and delivers to customers in fresh coolers; customers put them in the oven and have a meal within 30...
-
Hydrogen sulphide (a weak diprotic acid) is added to pure water at a concentration of 10- M. Assuming a temperature of 25 C and assuming ionic strength effects are negligible, determine the pH (to...
-
Gale Company has the following inventory and purchases during the fiscal year ended December 31, 2023. Beginning Inventory Feb. 10 purchased Feb. 20 sold Mar. 13 purchased Sept. 5 Oct. 10 purchased...
-
Constants for 16-18: Water: C = 4,180 J/kgoC, Lf = 334,000 J/kg, Lv = 2,260,000 Ice: C = 2000 J/kgoC Steam: C = 1996 J/kgoC In an experiment 2.5 kg of ice that is at -30 C is heated until it is water...
-
you think you are the strongest or most experienced when it comes to working with type? you want to improve when it comes to working with type? Are there areas within the subject that you do not...
-
Part 4: Review appraisal ABRG's Records Management Procedure requires client documents to be stored and maintained securely to ensure confidentiality. Electronic files must be saved as a zipped...
-
Show that the Lagrangian \(L=\left(q_{1} \dot{q}_{2}-q_{2} \dot{q}_{1} ight)^{2}-a\left(q_{1}-q_{2} ight)^{4}\) is invariant under the transformations and construct the corresponding conserved...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Borazine, B 3 N 3 H 6 is often referred to as inorganic benzene because of its similar structure. Like benzene, borazine has a delocalized system. Describe the molecular orbitals of the system....
-
Starting with benzene and the appropriate acyl chloride or acid anhydride, outline a synthesis of each of the following: (a) Butylbenzene (b) (c) (d) Diphenylmethane 9,10-Dihydroanthracene
-
Heating 1,1,1-triphenylmethanol with ethanol containing a trace of a strong acid causes the formation of 1-ethoxy-1,1,1-triphenylmethane. Write a plausible mechanism that accounts for the formation...
-
(a) Which of the following halides would you expect to be most reactive in an SN2 reaction? (b) In an SN1 reaction? Explain your answers. Br
-
A firm has 36.30 million shares of common stock outstanding at a price of $32.81 per share. The firm also has 492000.00 bonds outstanding with a current price of $1,089.00. The outstanding bonds have...
-
Commwest Bank owns $300 million of variable-rate notes yielding BBR plus 4 per cent. These loans are financed by $300 million of fixed-rate bonds costing 5 per cent.Northside Building Societyhas $300...
-
Mr. Smith enters into the long position in 5 S&P 500 futures contracts. His brokerage account requires an initial margin balance equal to 15% of the notional amount and a maintenance margin equal to...

Study smarter with the SolutionInn App


