Draw all lone pairs on each of the nitrogen atoms in the compounds below. First, review in
Question:
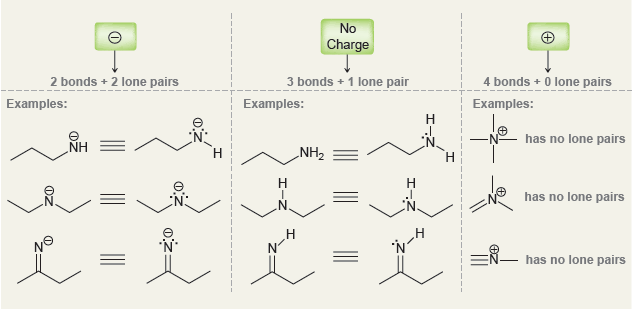
a.
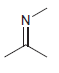
b.
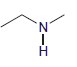
c.
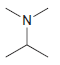
d.
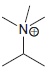
e.
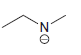
f.
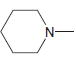
g.
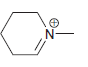
h.
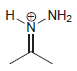
Transcribed Image Text:
No Charge 3 bonds + 1 lone pair 2 bonds + 2 lone pairs 4 bonds + 0 lone pairs Examples: Examples: Examples: н has no lone pairs NH NH2 has no lone pairs .N' has no lone pairs z:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
a b c d N...View the full answer

Answered By

Muhammad Khurram
I have strong General Management skills to apply in your projects. Over last 3 years, I have acquired great knowledge of Accounting, Auditing, Microsoft Excel, Microsoft PowerPoint, Finance, Microsoft Project, Taxation, Strategic Management, Human Resource, Financial Planning, Business Planning, Microsoft Word, International Business, Entrepreneurship, General Management, Business Mathematics, Advertising, Marketing, Supply Chain, and E-commerce. I can guarantee professional services with accuracy.
4.80+
249+ Reviews
407+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all lone pairs on each of the oxygen atoms in the compounds below. Before doing this, review in the following table, and then come back to these problems. Try to identify all lone pairs without...
-
The azide ion, N3-, is linear with two N-N bonds of equal length, 1.16 Ã. (a) Draw a Lewis structure for the azide ion. (b) With reference to Table 8.5, is the observed bond length consistent...
-
The following compound has three nitrogen atoms: Each of the nitrogen atoms exhibits a lone pair that can function as a base (to abstract a proton from an acid). Rank these three nitrogen atoms in...
-
If the owner of a company takes merchandise for personal use, what account is debited? a. Owners capital b. Owners withdrawals c. Purchases d. Cash
-
Per the Kiobel case, why might one disagree with the Court's Majority decision? Persuasively and rigorously explain.
-
Chrysler Group LLC. and Fiat SpA announced that they would partner to bring the subcompact Fiat 500 to be sold in the U.S. sometime in 2011. The Fiat 500 will be positioned as a direct competitor to...
-
A survey indicates that for each trip to a supermarket, a shopper spends an average of 43 minutes with a standard deviation of 12 minutes in the store. The lengths of time spent in the store are...
-
During 2013, Matti Conners, president of Maggert Company, was paid a semimonthly salary of $5,200. Compute the amount of FICA taxes that should be withheld fromher: OASDI HI a. 9th paycheck S b. 22nd...
-
Parkview Fish Tacos sells tacos for $4.80 each. The cost of each taco follows: Materials (food) Labor Variable overhead Fixed overhead ($7,200 per month, 4,500 tacos per month) Total costs per taco $...
-
Prepare a balance sheet as of June 30, for the J. L. Gregory Company, using the following data: Accounts payable Accounts receivable Accrued expenses Accumulated depreciation 241,000 505,000 107,000...
-
A carbene is a highly reactive intermediate in which a carbon atom bears a lone pair and no formal charge: How many hydrogen atoms are attached to the central carbon atom above?
-
Each of the following compounds contains both oxygen and nitrogen atoms. Identify all lone pairs in each of the following compounds: a. b. c. d. e. f. N. O=C=N
-
When 2-hepten-4-one is treated with LDA, a proton is removed from one of the gamma () positions. Identify which position is deprotonated, and explain why the proton is the most acidic proton in the...
-
_____________is a measure that may be used to evaluate the extent to which a company is controlling its operating costs or how efficiently the company is getting oil and gas out of the ground, or...
-
Explain how estimation of a companys net proved reserves differs when operating under a concessionary contract versus a PSC.
-
What is the term describing two tracts of land that share only a corner? a. Adjacent b. Contiguous c. Annexed d. Indexed e. None of these
-
Generally, percentage depletion is allowed for ___________. a. Royalty owners b. Integrated producers c. Independent producers d. All of these e. None of these
-
Describe the similarities and differences between PSCs and risk service contracts.
-
How do you create a new database in MySQL?
-
Find the APR in each of the following cases: NUMBER OF TIMES COMPOUNDED Semiannually Monthly Weekly Infinite EAR APR 10.4% 8.9 11.6 15.4
-
Propose structures for compounds E-H. Compound E has the molecular formula C5H8 and is optically active. On catalytic hydrogenation E yields F. Compound F has the molecular formula C5H10, is...
-
Consider the interconversion of cis-2-butene and trans-2-butene. (a) What is the value of Ho for the reaction cis-2-butene : trans-2-butene? (b) Assume Ho Go. What minimum value of DG would you...
-
(a) Partial dehydrohalogenation of either (1R, 2R)-1,2-dibromo-1,2-diphenylethane or (1S, 2S)-1,2-dibromo-1,2-diphenylethane enantiomers (or a race mate of the two) produces...
-
given l o g x 5 = l o g 5 x solve for x
-
Solve the formula 8 x 3 y = - 1 9 for y .
-
Q6 Find the total amount for of the current balance for each underwriter and find out top 10 richest underwriter Q7 Create a pivot table for the entire data and find out the total current balance for...

Study smarter with the SolutionInn App


