Draw all lone pairs on each of the oxygen atoms in the compounds below. Before doing this,
Question:
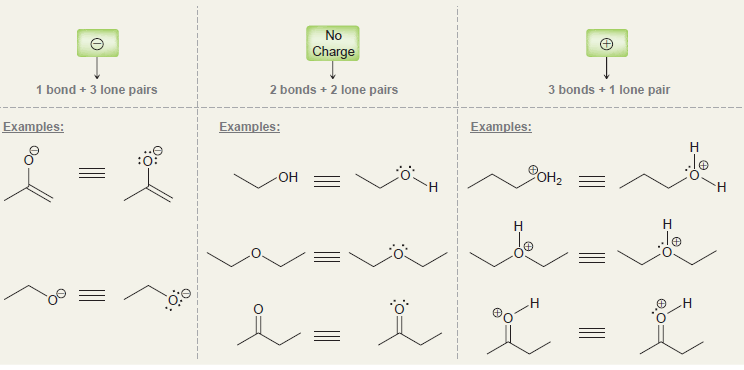
a.
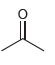
b.
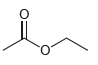
c.
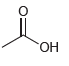
d.
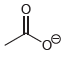
e.
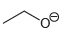
f.
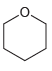
g.
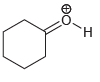
h.
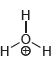
i.
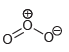
j.
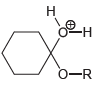
Transcribed Image Text:
No Charge 3 bonds + 1 lone pair 1 bond + 3 lone pairs 2 bonds + 2 lone pairs Examples: Examples: Examples: Н eона Он н Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a b ...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all lone pairs on each of the nitrogen atoms in the compounds below. First, review in the following table, and then come back to these problems. Try to identify all lone pairs without having to...
-
Sulfur dioxide (SO2) consists of two oxygen atoms (each of mass 16 u, where u is defined in Problem 39) and a single sulfur atom (of mass 32 u). The center to-center distance between the sulfur atom...
-
Each of the following compounds contains both oxygen and nitrogen atoms. Identify all lone pairs in each of the following compounds: a. b. c. d. e. f. N. O=C=N
-
At January 1, 2024, Mahmoud Industries, Inc., owed Second BancCorp $12 million under a 10% note due December 31, 2026. Interest was paid last on December 31, 2022. Mahmoud was experiencing severe...
-
On January 1, 20X1, Big Company (Big) bought 30% of the outstanding stock of Little Company (Little) for $110,000 which provided Big with the ability to significantly influence the decisions of...
-
Discuss the three dimensions of channel structure that should be considered in developing alternative channel structures.
-
P(x < 180) The random variable x is normally distributed with mean = 174 and standard deviation = 20. Find the indicated probability.
-
Grant Film Productions wishes to expand and has borrowed $100,000. As a condition for making this loan, the bank requires that the business maintain a current ratio of at least 1.50. Business has...
-
I've been tasked with applying Cost-Benefit Analysis (CBA), SWOT analysis, and decision tree analysis tools to the long-term decision of purchasing a car. I'm looking for insight to further my...
-
Your brother has just started a new job as the Controller of an IESBA restricted audit client. You do not serve on the audit engagement. What steps must you take to ensure your independence is not...
-
For each of the compounds below determine whether any of the oxygen atoms bear a formal charge: a. b. c. d. ::
-
A carbene is a highly reactive intermediate in which a carbon atom bears a lone pair and no formal charge: How many hydrogen atoms are attached to the central carbon atom above?
-
Find the present and future values of an income stream of $2000 per year for 15 years, assuming a 5% interest rate compounded continuously.
-
Support equipment and facilities that service a particular field or other area constituting a cost center should be capitalized and depreciated using the _________ method over the proved developed...
-
If the expected cash flow approach is used to estimate the fair value of an ARO, what discount rate should be used? a. Weighted average cost of capital b. Credit-adjusted risk-free rate c....
-
When are companies required to remeasure AROs at fair value? a. Annually b. When discount rates change c. When events and circumstances indicate that the underlying assets carrying value might not be...
-
When is a company required to test for impairment (according to ASC 360-10-35)? a. Annually b. When events or circumstances indicate that the assets carrying value may be recoverable c. When events...
-
How influential would you say that the factors noted in Figure 2.4 were in creating the drive to introduce SHRM in major organisations? Figure 2.4: Cultural strategies Corporate strategy HR...
-
What are the three MySQL programs that are recommended as a useful set of MySQL software products?
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
Provide a mechanistic explanation for each of the following reactions: (a) (b) (c) (d) acid (cat.) OH (major product) acid (cat.) (major product) AgNO3 EtOH (major product) Ph. H EtONa EtOH, ,Ph Ph...
-
Caryophyllene, a compound found in oil of cloves, has the molecular formula C15H24 and has no triple bonds. Reaction of caryophyllene with an excess of hydrogen in the presence of a platinum catalyst...
-
Squalene, an important intermediate in the biosynthesis of steroids, has the molecular formula C30H50 and has no triple bonds. (a) What is the index of hydrogen deficiency of squalene? (b) Squalene...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. Explain the statement, "all business is sales." What six things matter? If...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. No matter what you have done in the past, the boss really doesn't care. Why?...
-
Do you think Richard Scrushy should have been found guilty or acquitted for his role in the case against HealthSouth? https://www.wsj.com/articles/SB111702610398942860

Study smarter with the SolutionInn App


