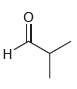
Draw both resonance structures of the enolate formed when each of the following ketones is treated with
Question:
(a)
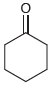
(b)
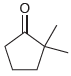
(c)
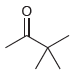
(d)
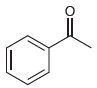
(e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When each of the following ketones is treated with aqueous sodium hydroxide, the aldol product is obtained in poor yields. In these cases, special distillation techniques are used to increase the...
-
When 2-methylcyclohexanone is treated with a strong base, two different enolates are formed. Draw both of them.
-
Draw the enolate ion that is formed when each of the following compounds is treated with sodium ethoxide. In each case, draw all resonance structures of the enolate ion, and predict whether a...
-
What are the advantages and disadvantages of seismic reflection data sets?
-
Who are the suppliers of rhino and what was the average selling price of a rhino? How does the sale of rhino help SANParks?
-
Liz Claiborne, Inc. (Claiborne) is a large maker of sportswear in the United States and a well known name in fashion, with sales of more than $ 1 billion per year. Claiborne distributes its products...
-
The built-up shaft consists of a pipe AB and solid rod BC. The pipe has an inner diameter of 25 mm and outer diameter of 30 mm. The rod has a diameter of 15 mm. Determine the average normal stress at...
-
Highlow methodmissing amounts. The following data have been extracted from the records of Puzzle, Inc.: Required: a. Calculate the missing costs. b. Calculate the cost formula for mixed cost using...
-
StorSmart Company makes plastic organizing bins. The company has the following inventory balances at the beginning and end of March: S Raw materials Work in process Finished goods Beginning Inventory...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Draw the two possible enols that can be formed from 3-methyl-2-butanone, and show a mechanism of formation ofeach under base-catalyzed conditions.
-
What is meant by queue discipline?
-
Why does the cutoff wavelength in Fig. 28-11 imply a photon nature for light?
-
What are the components of a BOM?
-
What are the two types of operational management of business processes?
-
Most entrepreneurs use their own savings or borrow from family and friends to launch a start-up. How much capital could you possibly raise?
-
What are the various types of BPM activities?
-
Miguel and Juliana Santiago, ages 30 and 28, were recently married in Dallas. Miguel is an electrical engineer with Silicon Systems, a computer component design firm. Juliana has a masters degree in...
-
Use the intermediate value theorem to show that each polynomial function has a real zero between the numbers given. (x) = x 4 - 2x 3 - 2x 2 - 18x + 5; 3.7 and 3.8
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) Benzyl methyl ether from toluene (b) (c) (d) CH2CH O from cyclopentene 7-CO.H...
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Eckert Company issues $10,000,000, 6%, 5-year bonds dated July 1, 2017 on July 1, 2017. The bonds pay interest semiannually on December 31 and June 30. The bonds are issued to yield 5%. What are the...
-
Spencer Company acquired 70% of the common stock of Dean Corporation on August 1, 2025. For 2025, Dean reported revenues of $960,000 and expenses of $780,000, all reflected evenly throughout the...
-
1. Solve the following systems of congruences or explain why they do not have solutions. In case there are solutions they must be given in the modulus that is the LCM of the congruence modulus. (a)...

Study smarter with the SolutionInn App


