Each of the following compounds contains two carbonyl groups. Identify which carbonyl group will exhibit a signal
Question:
Each of the following compounds contains two carbonyl groups. Identify which carbonyl group will exhibit a signal at lower wave number.
a.
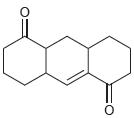
b.
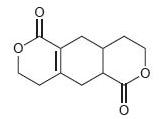
c.
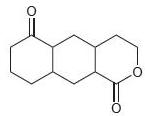
Transcribed Image Text:
O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a b c lowe...View the full answer

Answered By

Collins Omondi
I have been an academic and content writer for at least 6 years, working on different academic fields including accounting, political science, technology, law, and nursing in addition to those earlier listed under my education background.
I have a Bachelor’s degree in Commerce (Accounting option), and vast knowledge in various academic fields Finance, Economics, Marketing, Management, Social Science, Women and Gender, Business law, and Statistics among others.
4.80+
4+ Reviews
16+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following compounds is characterized by a 1H NMR spectrum that consists of two peaks, both singlets, having the chemical shifts indicated. Identify each compound. (a) C6H8; 2.7 ppm (4H)...
-
Each of the following compounds is unstable and either exists as an isomer or spontaneously decomposes to other compounds. In each case, give the more stable isomer or decomposition product and...
-
Each of the following compounds exists as a fluxional molecule that is interconverted into one or more identical forms by the sigmatropic process indicated. Draw one structure in each case that...
-
A. Discuss the following conceptual model/framework of Orem's Self-Care Theory CONDITIONING FACTORS (12 MARKS) Orem's Self-Care Theory Conceptual Framework SELF-CARE AGENCY SELF-CARE DEFICIT NURSING...
-
What are some similarities in the world's financial systems?
-
Until the discoveries of Werner, it was thought that the presence of carbon in a compound was required for it to be optically active. Werner prepared the following compound containing OH- ions as...
-
At the beginning of the year 2013, Company XYZ had an inventory of 8,000 widgets with a unit cost of \(\$ 6.00\). During that year, the following purchases of widgets were made: At the end of 2013,...
-
The condenser of a steam power plant consists of AISI 302 stainless steel tubes (k s = 15 W/m K) each of outer and inner diameters Do = 30 mm and Di = 26 mm, respectively. Saturated steam at 0.135...
-
Four grams of musk oil are required for each bottle of Mink Caress, a popular perfume made by a company in western Siberia. The cost of the musk oil is $1.80 per gram. Budgeted quarterly production...
-
It's amazing how much difference there is in the way proposals are presented at two different firms," said John Woods to his assistant, Pete Madsen, as he pointed to the stack of capital investment...
-
What are the main categories of government spending? What are the main categories of government revenue?
-
Eric and Kyle are fishermen with different equipment and, as a result, different costs for catching fish. Erics costs for catching fish are $1,000 per ton for the first five tons and then $2,500 per...
-
What determines the quality of earnings?
-
Do you think Starbucks could have benefited from placing a greater reliance on franchising over the years? What are the benefits to Starbucks as a result of the decision to avoid going the franchise...
-
A common-size income statement for Santa Enterprises 2017 operations follows. Using the firms 2017 income statement, develop the 2017 common-size income statement and compare it with the 2016...
-
What are the main reasons that many U.S. franchise systems are expanding into global markets? Do you think this expansion will continue to gain momentum or will decline over time? Provide an example...
-
To date, every court case that has been adjudicated in the United States indicates that franchisors do not have a fiduciary responsibility to their franchisees. What do these rulings suggest to...
-
Reread the Opening Profile, which focuses on Joseph Keeley and the franchise organization that he foundedCollege Nannies & Tutors. Think of an activity, other than nanny and tutoring services, that...
-
Johnston Company (JC) sells two types of bicycles with details as follows for 2019: Required: 1. Calculate the budgeted contribution margin for each model. 2. Calculate the following variances: a....
-
The outer loop controls the number of students. Note that the inner loop of this program is always executed exactly three times, once for each day of the long weekend. Modify the code so that the...
-
Treatment of the following alkene with a peroxyacid yields an epoxide different from that obtained by reaction with aqueous Br2 followed by base treatment. Propose structures for the two epoxides,...
-
Draw structures corresponding to the following IUPAC names: (a) Ethyl 1-ethylpropyl ether (b) Di (p-chlorophenyl) ether (c) 3, 4-Dimethoxybenzoic acid (d) Cyclopentyloxycyclohexanc (e)...
-
Give IUPAC names for the followingstructures: (c) CH (b) (a) " SH (f) CH (e) (d) CHCH0- CHH 5. NO2 SCH3 SCH3 (i) (h) CH CH CH3CH2CHCHCHSCHCH3 CH CH (g) CCH H
-
What choice is a multiple of 5? a) 93 b) 63 c) 65 d) 84
-
Determine how many tablets will be needed to give the dosage. Prepare a dosage of 6.4 mg using tablets with a strength of 1.6 mg.
-
(a). Company Inc., private consumer regulator firm, claims that the standard deviation of the rating of Formula One races in the town main car racing circuit was at least 15.34. (b). An outside...

Study smarter with the SolutionInn App


