Each of the following compounds possesses a plane of symmetry. Find the plane of symmetry in each
Question:
a.
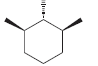
b.
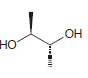
c.
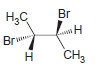
d.
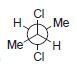
Transcribed Image Text:
но Но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
a...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
To which compound class does each of the following compounds belong? (a) (b) C=N
-
Each of the following molecules has one plane of symmetry. Find the plane of symmetry in each case: a. b. c. d. e. f. CI Me Me Br
-
Each of the following compounds possesses carbon atoms that are chirality centers. Locate each of these chirality centers, and identify the configuration of each one: a. b. c. d. e. f. Ephedrine A...
-
Exercises 11-16: For the measured quantity, state the set of numbers that most appropriately describes it. Choose from the natural numbers, integers, and rational numbers. Explain your answer....
-
In terms of strategic management, how does a new venture's situation differ from that of an ongoing small company? How should a small company engage in environmental scanning? To what aspects of the...
-
A firm wants to bid on a contract worth \(\$ 80,000\). If it spends \(\$ 5000\) on the proposal it has a \(50-50\) chance of getting the contract. If it spends \(\$ 10,000\) on the proposal it has a...
-
Analyze the chemical process data in Table B. 5 for evidence of multicollinearity. Use the variance inflation factors and the condition number of \(\mathbf{X}^{\prime} \mathbf{X}\). Run No. y X x2 X3...
-
How much of the following expenses are currently deductible by a cash basis taxpayer? a. Medical prescriptions costing $20 paid by credit card (medical expenses already exceed the 10% of AGI floor)....
-
Maslow's hierarchy of needs provide a useful framework for understanding how and why local products and brands can be extended beyond home-country borders. How can Maslow's needs hierarchy be used in...
-
1. Identify at least one important hub and one important broker in the Unit 8200 network. 2. If you were a foreign cyber-security company, what would you do to access Israels expertise?
-
(R)-Limonene is found in many citrus fruits, including oranges and lemons: For each of the following compounds identify whether it is (R)-limonene or its enantiomer, (S)-limonene: a. b. c. d.
-
For each of the following pairs of compounds, determine the relationship between the two compounds. a. b. c. d. e. f. g. h. i. j. k. l. Me Me -CI CI- - - - - Me Me
-
Again consider the car sales rep in question 67. What is the probability that she will make at least two sales after speaking to four customers? Question 67 A car sales representative sees an average...
-
If the Fed had adopted inflation targeting and used the Taylor rule to hit its target, how would the course of the federal funds rate have been different from the course that it did take? How would...
-
Set up the equilibrium equations required to analyze the frame in Figure P10.33 by the slopedeflection method. Express the equilibrium equations in terms of the appropriate displacements. EI is...
-
What are the methods of owning property with others?
-
One of the strategies that has been recommended for enhancing job satisfaction is to make jobs more fun. We all like having fun, of course, but do you really think these matters when it comes to job...
-
Japans Gross Domestic Product for the Third Quarter of 2017 Japans GDP rose by 0.3 percent in the third quarter, primarily because of an increase in exports and a slow increase in investment....
-
We know that speed v = distance/time. Show that when the distance traveled is one wavelength and the time of travel is the period T (which equals 1/frequency) you get v = f.
-
Bonus shares can be issued out of revenue reserves. True/False?
-
Allowing acetone to react with 2 molar equivalents of benzaldehyde in the presence of KOH in ethanol leads to the formation of compound X. The 13C NMR spectrum of X is given in Fig. 19.1. Propose a...
-
Provide a mechanism for each of the following reactions. (a) (b) 0O (1) NaOEt, EtOH (1) NaOEt, EtOH (2) H3o
-
(a) Deduce the structure of product A, which is highly symmetrical: The following are selected spectral data for A: MS (m/z): 220 (M+.) IR (cm-1): 2930, 2860, 1715 1H NMR (δ): 1.25 (m),...
-
I. Analyze the geometric construction of the systems shown as in figures. A Figure 1-1 E Figure 1-2 Figure 1-3 bisg ela ad bloode vuilidizel Figure 1-4
-
structural engineering course, i need it asap please. thanks A. Calculate the reactions of the two-span frame shown in figure Figure 4-1 2.5kN/m (a) D E F A B 2m 2m 4m 0 4m B. Calculate member-end...
-
As a hired structural engineering consultant, you were requested to evaluate a four-story building that is damaged by a Magnitude 6 earthquake. The client has no background in any civil/structural...

Study smarter with the SolutionInn App


