Explain the vast difference in pK a values for the following two apparently similar compounds. pk, =
Question:
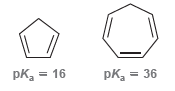
Transcribed Image Text:
pk, = 16 pk, = 36
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Cyclopentadiene is more acidic because its conjugate base is hig...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain each difference in reactivity toward nucleophiles. a. Esters are less reactive than ketones. b. Benzoyl chloride is less reactive than cyclohexanecarbonyl chloride.
-
Explain the difference in the melting points of the following compounds: Only one of the two can form intramolecular hydrogen bonds? NO2 NO OH OH m.p. 115C m.p. 45C
-
Explain how you could distinguish between the following two compounds using NMR coupling constants. (These compounds are derived from glucose, by a reaction we shall study in Chapters 16 and 22.) A B...
-
For the transfer function below, find the constraints on K 1 and K 2 such that the function will have only two j poles. K1s + K2 T(s) = s4 + K1s + s? + K2s + 1
-
Please calculate CAPM of Asset J with the following information: where, kj = required return on asset j, Rf = risk-free rate of return, (6%) bj = beta coefficient for asset j, (1.75) Rm = market...
-
For the same potential as above, \(V(r)=V_{0} /\left(r^{2}+a^{2} ight)\), calculate \(\Delta(b)\) for the eikonal approximation. If the potential turns off \((V(r)=0)\) for \(r \geq r_{0}\),...
-
For each of the following situations, calculate a \(95 \%\) confidence interval for the mean ( \(\sigma\) known), beginning with the step, "Identify the critical value of \(z\)." X 50.00, X = 3.00 X...
-
1. What were Price Waterhouses fatal flaws? 2. Does Hopkinss treatment here make good business sense? Explain. 3. How would you avoid the problems in this case? In a jointly prepared statement...
-
On June 3 0 , Indigo Corporation discontinued its operations in Mexico. During the year, the operating income was $ 2 1 0 , 0 0 0 before taxes. On September 1 , Indigo disposed of the Mexico facility...
-
Presented below is a combined single-step income and retained earnings statement for Hardrock Mining Co. for 20X1. Statement of Income and Retained Earnings for the Year Ended December 31, 20X1 ($ in...
-
Determine whether each of following ions is aromatic, nonaromatic, or antiaromatic. a. b. c. d. 0:
-
Predict which compound will react more readily in an S N 1 process, and explain your choice. Br -Br
-
The wavelengths of the yellow doublet components of the resonance Na line caused by the transition 3P 3S are equal to 589.00 and 589.56 nm. Find the splitting of the 3P term in eV units.
-
During June, Home Hardware made sales of $ 49700 and ended the month with inventories totaling $9600. Cost of goods sold was $ 19800. Total operating expenses were $ 6600.How much net income did Home...
-
Assume that the market equilibrium fee of physician consultation services is $150. Using demand-supply curve of physician services, show the changes in market equilibrium if all individuals in the...
-
Revenue $525,000 Sales Returns $13,900 Sales Discounts $12,400 Net Sales $498,700 Cost of Goods Sold $79,500 Gross Profit Gross Profit Margin % 84.06% Given the information above, what is the Gross...
-
If GDP was $3 trillion over a certain time period and aggregate expenditure was $2.8 trillion over the same time period, what should be expected to occur given that the country does not export?...
-
Consider a perfectly competitive firm with the following cost function: TC = 2(Q) + 1600 The firm sells its output for $168 per unit. A. B. C. D. Find the profit-maximizing level of output. Represent...
-
Assume that the spinner cannot land on a line. Determine the probability that the spinner lands on (a) Red. (b) Green. (c) Yellow. (d) Not yellow.
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
Write conformational structures for all of the stereoisomers of 1,3-diethylcyclohexane. Label pairs of enantiomers and meso compounds if they exist.
-
Tartaric acid [HO2CCH(OH)CH(OH)CO2H] was an important compound in the history of stereochemistry. Two naturally occurring forms of tartaric acid are optically inactive. One optically inactive form...
-
(a) An aqueous solution of pure stereoisomer X of concentration 0.10 g mL-1 had an observed rotation of -300 in a 1.0-dm tube at 589.6 nm (the sodium D line) and 25oC. What do you calculate its [a]D...
-
calculate the payback period, internal rate of return, and net present value of the proposed mine
-
You need to choose between the following two design projects: year Cash flow A cash flow B 0 -$81,000 -$21,800 1 38,600 12,200 2 38,600 12,200 3 38,600 12,200 a) If the required return is 10 percent...
-
Home Furnish has bonds on the market with 13 years to maturity and a yield to maturity of 8.5.In the financial newspaper, Home Furnish bonds is quoted as 127.223% of par value. The bonds make...

Study smarter with the SolutionInn App


