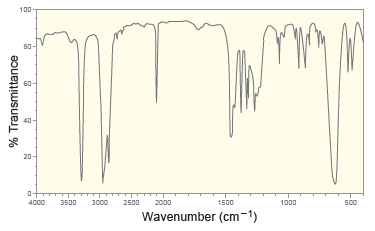
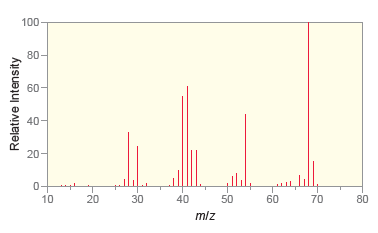
Following are the IR spectrum and mass spectrum of an unknown compound. Propose at least two possible
Question:
Transcribed Image Text:
100 80 20 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) % Transmittance 100 80 - 60- 40- 20- 10 20 30 40 50 60 70 80 mlz m/z Relative Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The IR spectrum indicates the presence of a tri...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose at least two different structures for a compound with six carbon atoms that exhibits the following features: a. All six carbon atoms are sp 2 hybridized. b. Only one carbon atom is sp...
-
Propose at least two strategies to avoid assumptions in a multiyear plan. Justify your response. Recommend at least two best practices for analyzing multiyear financial statements. Justify your...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
In Exercises 8586, find a. (f g)(x); b. (g f)(x); c. (f g)(3). f(x)=x, g(x) = x + 1
-
Record the current price of the stock for each company you selected in Week 3's Stock Journal. Using MS Excel spreadsheet or MS Word document put stock prices side-by-side, to show your comparison....
-
An analyst is reviewing the valuation of DuPont (NYSE: DD) as of the beginning of July 2013 when DD is selling for \($52.72\) . In the previous year, DuPont paid a \($1.70\) dividend that the analyst...
-
Upon reviewing recent use of conference rooms at an engineering consulting firm, an industrial engineer determined the following probability distribution for the number of requests for a conference...
-
On July 1, 2017, Lula Plume created a new self-storage business, Safe Storage Co. The following transactions occurred during the company's first month. July 1 Plume invested $30,000 cash and...
-
How do charismatic leadership attributes influence organizational culture and employee motivation, and what potential pitfalls should leaders be mindful of when exercising charismatic influence ?...
-
In a certain city, 30% of the families have a MasterCard, 20% have an American Express card, and 25% have a Visa card. Eight percent of the families have both a MasterCard and an American Express...
-
Following are the IR spectrum and mass spectrum of an unknown compound. Propose at least two possible structures for the unknown compound. 100- 80- 40- 20- 4000 3500 3000 2500 2000 1500 1000 500...
-
Consider the following sequence of reactions: a) Explain how you could use IR spectroscopy to differentiate between compounds F and G. b) Explain how you could use IR spectroscopy to differentiate...
-
How would you determine the MIME type of a file?
-
Explain the process of portfolio management and its value.
-
Determine the approximate values of force in each member of the truss in Figure P13.13. Assume that the diagonals can carry either tension or compression force. 20 kips 15' A 20' C B 20 kips
-
The facility to place marketing content online in a variety of forms and on numerous sites means that potential and existing customers have a wealth of sources and material with which to inform...
-
Identify the similarities in Figures 3.8 (a) and 3.10. Identify the differences. Figure 3.8a Figure 3.10 Output variety High Much Low None Project None One Job Cell Few Flow Many Batch size (a) None...
-
Is the stretched-S life-cycle project form more common or the exponential form? What other aspects of managing a project are affected by the nature of the project form besides budget- ing and early...
-
Suppose that the mass of the proton and the mass of the neutron were each exactly 1.0 u. What would the mass defect for a copper-63 nucleus be under these circumstances if its atomic mass is 62.5 u?
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
Draw a structural formula for each of the following compounds. (Several formulas may be possible in each case.) An alcohol with the molecular formula C5H10O
-
A certain compound was found to have. the molecular formula C5H12O2. To which of the following compound classes could the compound belong? Give one example for each positive answer, and explain any...
-
Draw the structures and give the names of all isomers of octane with Six carbons in their principal chains.
-
A production department reports the following conversion costs. Equivalent units of production for conversion total 436,000 units this period. Calculate the cost per equivalent unit of production for...
-
If you were asked whether a large university such as Tennessee or Michigan with a large seating capacity for their football stadiums should build a new football stadium, how would you respond and...
-
J is going to receive a 30-year annuity of 8,500 and L is going to receive perpetuity of 8,500. If the appropriate interest rate is 6%, how much more is L's cash flow worth?

Study smarter with the SolutionInn App


