For each of the compounds below, locate the pattern we just learned (lone pair next to a
Question:
a.
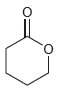
b.
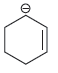
c.
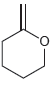
d.
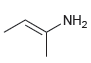
e.
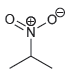
f.
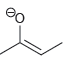
g.
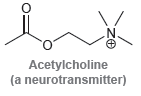
h.
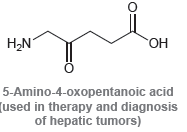
Transcribed Image Text:
NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the compounds below determine whether any of the nitrogen atoms bear a formal charge: a. b. c. d. N.
-
For each of the compounds below determine whether any of the oxygen atoms bear a formal charge: a. b. c. d. ::
-
For each of the compounds below, locate the lone pair adjacent to a positive charge and draw the resonance structure: a. b. c. N.
-
Mixing together solutions of acetic acid and sodium hydroxide can make a buffered solution. Explain. How does the amount of each solution added change the effectiveness of the buffer?
-
Ozark Corporation reported net income of $100,000 for 20X5. The income statement revealed sales of $1,000,000; gross profit of $520,000; selling and administrative costs of $340,000; interest expense...
-
When the Gallup Organization conducted the poll introduced in Example 1, 54.9% of those surveyed considered the amount of federal income tax they have to pay as fair. Gallup reported its survey...
-
For each of the following, (a) calculate the mean, standard deviation, and standard error of the mean for each Group and (b) construct a bar graph (assume the independent variable is condition and...
-
Starwood Hotels (Starwood) owns and operates many hotel properties under well-known brand names, including Sheraton, W, Westin, and St. Regis. Starwood focuses on the upper end of the lodging...
-
The balance sheet for Trinity Motors Company for December 3 1 of 2 0 2 2 and 2 0 2 3 . What was the cash flow to stockholders in 2 0 2 3 ?
-
A food company manufactures five types of 8 oz Trail mix packages using different mixtures of peanuts, almonds, walnuts, raisins, and M&Ms. The mixtures have the following compositions: How many...
-
An ideal gas undergoes a single-stage expansion against a constant external pressure P external = P f at constant temperature from T,P i ,V i , to T,P f ,V f . a. What is the largest mass m that can...
-
Draw the resonance structure(s) for each of the compounds below: a. b. c. d.
-
Use a graphing utility to graph any five of the ellipses that you graphed by hand in Exercises 118. Data from exercise 1-18 1. 2. 3. 4. 5. (-4,0)- F YA 5(0,2) 1 + 16 (4,0) X5 x 12 (0, -2) 2 = 1 4
-
Use the PV=FV/(1+r)t formula and solve for r to get the geometric average increase in dividends. So, .08 = .28/(1+r)10
-
How measure of variation (CV or standard deviation) is better to use when comparing Verizon data speeds in Mbps and magnitudes of earthquakes in RM (Richter magnitude)?
-
What is FTX? How does it operate? What factors triggered its bankruptcy? What are the implications for cryptocurrencies at large?
-
Tel Tec has just announced that their next dividend will be $2.25. In the announcement, management has projected that earnings will grow at 10 percent per year for the following 3 years, then slow to...
-
Discuss the role of post-translational modifications, such as glycosylation and phosphorylation, in regulating protein function and cellular signaling pathways within the context of intracellular...
-
If Nature suddenly changed and Plancks constant became a much larger number, what effect would this have on things such as solar cells, atomic emission and absorption spectra, lasers, and so on?
-
Privitera and Freeman (2012) constructed a scale to measure or estimate the daily fat intake of participants; the scale was called the estimated daily intake scale for fat (EDIS-F). To validate the...
-
Write structures for the major organic products from the following reactions. Show stereoisomers where applicable. (a) (b) (c) (d) (e) H20, H.So4 dr (1) BH THF (2) NaOH, H20, Br (1) 03 (2) Meps
-
Give the structure of the products that you would expect from the reaction of 1-butyne with: (a) One molar equivalent of Br2 (b) One molar equivalent of HBr (c) Two molar equivalents of HBr (d) H2...
-
Provide mechanistic explanations for the following observations: (a) (b) HCI CI CI Cl CI HCI
-
Work out which number line below shows the values that x can take if 3 x 2. - A +12345678910 8 9 10 B D 5 6 7 8 9 10 6 7 8 E 2 F 3 4 7 8 9 10 1 2 3 4 5 6 7 8 9 10
-
Application Case Coffee Shop is a company dedicated to the commercialization of "signature coffee", a category of high quality coffee that is grown, processed and prepared with a high degree of...
-
The issuing company Zapatos y Bolsas Veloz, SAB de C.V., dedicated to the manufacture and sale of vinyl leather bags for more than 25 years under the Bolsas Finas brand, the company obtained in the...

Study smarter with the SolutionInn App


