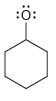
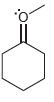
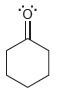
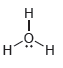
For each of the compounds below determine whether any of the oxygen atoms bear a formal charge:
Question:
a.
b.
c.
d.
Transcribed Image Text:
:ö:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a b ...View the full answer

Answered By

Amit Choudhary
I'm new in this profession regarding online teaching but previously i used to teach students near my college. I am teaching on online platform since last year and got good support from the students. I'm teaching on platforms like chegg and vedantu and also at my home in free time.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the compounds below determine whether any of the nitrogen atoms bear a formal charge: a. b. c. d. N.
-
For each of the compounds below, locate the pattern we just learned (lone pair next to a Ï bond) and draw the appropriate resonance structure: a. b. c. d. e. f. g. h. NH2
-
For each of the compounds below, locate the lone pair adjacent to a positive charge and draw the resonance structure: a. b. c. N.
-
Which of the units listed in Multiple-Choice Question 9 are appropriate for the biologically equivalent dose that results when a person is exposed to radiation? Data From Question 9 Which of these...
-
On January 1, 20Y1, Martin Manufacturing paid cash for a new piece of manufacturing equipment. The machine cost $40,000 and had an estimated useful life of 5 years with a $5,000 salvage value ....
-
Show that the factor \(\left(z_{i}-z_{j} ight)^{m}\) in the Moore-Read wave function (also present in the phenomenological "Laughlin wave function" for a theoretical description of the FQHE) implies...
-
You have collected the following data: If you randomly select one of these 10 numbers, what is the probability the number (X) will be ... a. equal to 4 ? b. equal to 7 ? c. less than 5 ? d. greater...
-
Suppose the interest rate (on an annual basis) on three-month Treasury bills is ten percent in London and six percent in New York, and the spot rate of the pound is $2. a. How can a U.S. investor...
-
If two trading partners suddenly become entangled in a trade war, what impact will this have on each nation? Explain.
-
Determine the Miller indices for the planes shown in the following unitcell: +y B.
-
Atenolol and enalapril are drugs used in the treatment of heart disease. Both of these drugs lower blood pressure (albeit in different ways) and reduce the risk of heart attack. Using the following...
-
Draw all lone pairs on each of the oxygen atoms in the compounds below. Before doing this, review in the following table, and then come back to these problems. Try to identify all lone pairs without...
-
What is an NGO?
-
The company has provisioned $US1.7 billion ($2.4 billion) to fund Renova's work to December. Discuss the difference between provision and contingent liability from an accounting perspective.
-
Tweaks for newsletter New ideas for newsletter add on. For company newsletter not for email newsletter How can we implement it in a company of solar power energy?
-
Newsletters, especially virtual newsletters, are an essential way for teachers to communicate with parents of children in their classroom on a regular basis.Newsletters can be sent monthly to keep...
-
A lightning bolt strikes a tree, moving 16.97 C of charge through a potential difference of 140 MV. What energy was dissipated? 1MV = 106V You need to express you answer in MJ: 1 MJ = 10J You should...
-
Estimate the limit 4 -1 lim- x0 cosx1
-
Fill in the blank with an appropriate word, phrase, or symbol(s). Probability determined by the relative frequency of occurrence of an event, or actual observations of an experiment is called...
-
Define the term utility software and give two examples.
-
Give the products that would be formed when each of the following alcohols is subjected to acid-catalyzed dehydration. If more than one product would be formed, designate the alkene that would be the...
-
1-Bromobicyclo [2.2.1] heptane does not undergo elimination (below) when heated with a base. Explain this failure to react. (Construction of molecular models may help.) Br
-
When the deuterium-labeled compound shown at right is subjected to dehydrohalogenation using sodium ethoxide in ethanol, the only alkene product is 3-methylcyclohexene. (The product contains no...
-
The balance sheet of Consolidated Paper, Incorporated, included the following shareholders equity accounts at December 3 1 , 2 0 2 3 : Paid - in capital: Preferred stock, 8 . 8 % , 8 7 , 0 0 0 shares...
-
Bangles Corporation issued 5-year, 11% bonds with a face value of $300,000 on April 1, 2019. Assume that the bonds are sold for $318,000. Interest is paid semiannually at October 1 and April 1....
-
What role do sensitivity analyses and scenario planning play in mitigating risk and assessing the resilience of financial projections to potential market fluctuations and macroeconomic variables?

Study smarter with the SolutionInn App


