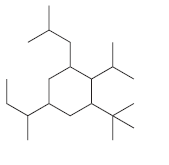
For each of the following compounds, identify all groups that would be considered substituents, and then indicate
Question:
(a)
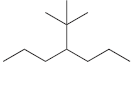
(b)
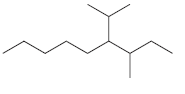
(c)
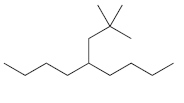
(d)
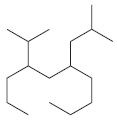
(e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b c d e S...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name each of the following compounds: (a) (b) (c) (d) (e) CH,CH NO2 di NO, CH2CH CI HO CH, OCH3 CH3 H3C CH(CH3)2 Cl 04 OCCCl3 Cl
-
Identify the common name for each of the following compounds: a. b. c. d.
-
Identify the functional groups in each of the following compounds: CH2CH2CH2CH2CH2CH3 Cl OH CH2CH2CH CH H2C CH3CH2CH2CH2 CH2CH2CH2CH3
-
Jillian operates a sporting goods store in a rented location at a shopping mall. She is insured under a CGL policy with the following limits: Indicate the dollar amount, if any, that Jillians insurer...
-
Watching an organization's failures play out on the news because they don't have a functioning crisis communications plan is both devastating and humiliating. Few things in corporate communications...
-
a. With what speed are pollen grains ejected from a bunch- berry flower? See Problem 47 for information. b. Suppose that 1000 ejected pollen grains slam into the abdomen of a \(5.0 \mathrm{~g}\) bee...
-
Consider the simple linear regression model fit to the steam plant data in Problem 2.12. Problem 2.12 The number of pounds of steam used per month at a plant is thought to be related to the average...
-
Teresas manufacturing plant is destroyed by fire. The plant has an adjusted basis of $260,000, and Teresa receives insurance proceeds of $400,000 for the loss. Teresa reinvests $425,000 in a...
-
The total factory overhead for Big Light Company is budgeted for the year at $739,820. Big Light manufactures two different products: night lights and desk lamps. Night lights are budgeted for 19,700...
-
Your brother has just started a new job as the Controller of an IESBA restricted audit client. You do not serve on the audit engagement. What steps must you take to ensure your independence is not...
-
Derive the following expression for calculating the isothermal change in the constant volume heat capacity: (CV/V)T = T (2P/T2)V.
-
The following substituent is called a phenyl group: With this in mind, identify the systematic name for each substituent below: Phenyl
-
Tell which line through the given points is steeper. Line 1: (-4, 2), (-3, 6) Line 2: (1, 6), (3, 8)
-
Lori is a student who teaches golf on Saturdays. In a year, she earns $20,000 after paying her taxes. At the beginning of 2021, Lori owned $1,000 worth of books, clothes, and golf clubs and she had...
-
Older Workers Are Being Pushed Out of the Job Market Compared to pre-pandemic levels, the number of people aged 55 and over who are in the labor force has decreased by 2 million. Corporate trends...
-
What was Michaels gross investment, depreciation, and net investment during 2022? Michael, an Internet service provider, bought an existing business worth $400,000 on December 31, 2021. During 2022,...
-
What is the principle of diminishing marginal benefit? In Problem 14, which marginal benefit diminishes fastest: Sams, Nicks, or the societys? Problem 14 The table sets out the marginal benefits that...
-
Calculate the inflation rates for the years ended June 2020 and June 2021. How did the inflation rate change in 2021? The BLS reported the following CPI data: June 2019 June 2020 June 2021 256.1...
-
If f() = tan = 3, find f( + ).
-
Using thermodynamic data from Appendix 4, calculate G at 258C for the process: 2SO 2 (g) + O 2 (g) 88n 2SO 3 (g) where all gases are at 1.00 atm pressure. Also calculate DG8 at 258C for this same...
-
Complete the molecular orbital description for the ground state of cyclopentadiene shown at right. Shade the appropriate lobes to indicate phase signs in each molecular orbital according to...
-
Rank the following dienes in order of increasing reactivity in a Diels-Alder reaction (1 = least reactive, 4 = most reactive). Briefly explain your ranking.
-
From each set of resonance structures that follow, designate the one that would contribute most to the hybrid and explain your choice: (a) (b) (c) (d) (e) (f) HO HO
-
Suppose that the net income view were to be correct in Problem 1. Describe a profitable investment strategy for investors in the companys bonds and equity if the company were to reduce its debt. What...
-
A piece of land in Ottawa with an area of 0.5 square kilometers is priced at 5200 Canadian dollars. If there are 0.9955 Canadian dollars per (U.S.) dollar, then what is the price in dollars per...
-
Marc and Mikkel are married and file a joint tax return. Marc and Mikkel earned salaries this year of $ 6 6 , 2 0 0 and $ 2 5 , 2 0 0 , respectively. In addition to their salaries, they received...

Study smarter with the SolutionInn App


