For each of the following IR spectra, determine whether it is consistent with the structure of a
Question:
a.
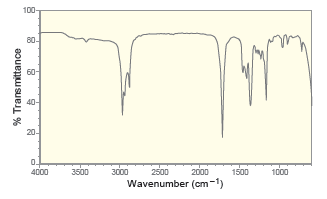
b.
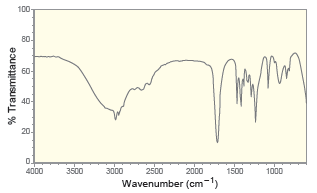
c.
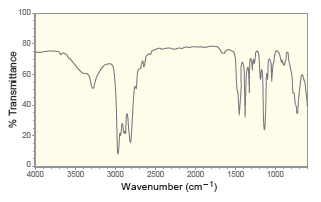
d.
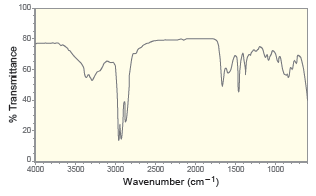
e.

f.
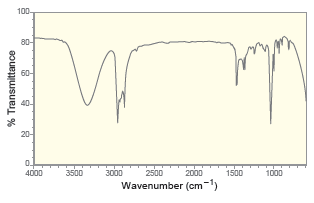
Transcribed Image Text:
100- 80- 60- 20- 0. 2500 Wavenumber (cm-) 4000 3500 3000 2000 1500 1000 % Transmittance 100 80- 60- 40- 20- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-') % Transmittance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a Ketone b...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following IR spectra, identify whether it is consistent with the structure of an alcohol, a carboxylic acid, or neither. a. b. c. d. e. f. 100- 80- 60- 40 20- 0. 2000 4000 3500 3000...
-
Determine the structure of each of the following compounds based on its mass, IR, and 1H NMR spectra. a. b. 100 u 50 27 114 20 40 60 80 100 120 m/z 3 2 2 28 2.9 10 13 14 15 6 6 5 2 (ppm) frequency...
-
An unknown compound gives the following mass, IR, and NMR spectra. Propose a structure, and show how it is consistent with the spectra. Show the fragmentations that give the prominent peaks at m/z...
-
ABC Company acquired 60,000 shares of DEF Incorporated's 200,000 ordinary shares on January 2, 2019 for P 4,000,000 when the net assets of DEF Incorporated was P 12,000,000. The excess of acquisition...
-
Using aggregate demand and supply, discuss an adverse oil shock, i.e., shortage of oil.
-
An unknown element is a nonmetal and has a valence electron configuration of ns2np4. a. How many valence electrons does this element have? b. What are some possible identities for this element? c....
-
Refer to Exercise 13.10 where the response is \(y=\) increase in particle size. Besides the first replicate, the investigators also performed the experiments that form the star part of the design....
-
Cummings Inc. had the following reconciliation at December 31, 2017: Fair value of plan assets.......................$5,000 PBO...............................................4,200 Funded...
-
Keller Company makes two models of battery-operated boats, the Sandy Beach and the Rocky River. Basic production information follows: Sandy Beach Direct materials cost per unit Direct labor cost per...
-
Many organizations use the same process capability standard for all their products or services (e. g., 1.33), but some companies use multiple standards: different standards for different products or...
-
The typical problem for generating parity in sports leagues is that a. There is no mechanism for bringing in new talent in a way that helps the bad teams. b. There is no means by which players on one...
-
Suppose that the government has been supporting the price of corn. Its free market price is $2.50 per bushel, but the government has been setting a support price of $3.50 per bushel. Which of the...
-
Ryan company paid salaries for the month amounting to $120,000. Of this amount, $30,000 was received by employees who had already been paid the $53,400 maximum amount of annual earnings taxable in...
-
Prepare a Statement of Revenues, Expenditures, and Changes in Fund Balance for the Broadus County Courthouse Bonds Debt Service Fund for 20X6, given the following information: The next debt service...
-
Laperla County entered into a capital lease on June 30, 20X8, for equipment to be used by General Fund departments. The capitalizable cost of the leased asset was $200,000. An initial payment of...
-
The school district ordered the following: (a) Record the following transactions in the General Fund General Ledger of Meadors Township using the consumption method (periodic inventory system) to...
-
Pick a topic in the general area of thermal energy systems of interest to you and research the ethical issues associated with environmental impact and/or sustainability related to subject you...
-
Conduct a parametric study of the economic selection problem given in Problem 2.29. Plot the annual cost of systems A, B, and C as a function of the MARR for the range 0 MARR 25%. Discuss the...
-
Cardinal Company is considering a five-year project that would require a $2,975,000 investment in equipment with a useful life of five years and no salvage value . The company's discount rate is 14%....
-
Ask students to outline the reasons why the various elements of culture (social structures and control systems, language and aesthetics, religion and other belief systems, educational systems, etc.)...
-
Identify the reagents represented by the letters a?c in the following scheme: Br Br Br Br
-
Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The PKa of un-substituted phenol, for example, is 9.89, while that of p-nitro...
-
Would you expect p-methyl phenol to be more acidic or less acidic than un-substituted phenol? Explain.
-
An automobile cooling system holds 16 L of water. How much heat does it absorb if its temperature rises from 20 C to 80 C? A 5.0 kg lead ball is dropped into a 2.0 L insulated pail of water initially...
-
A 15.0 kg box is allowed to slide from rest down a ramp that makes an angle of 30.0 o to the horizontal. The coefficient of friction between the box and the ramp is 0.400 and the ramp is 5.00 m long....
-
1. A bowling ball rolls without slipping up a ramp that slopes upward at an angle beta to the horizontal. Treat the ball as a uniform, solid sphere, ignoring the finger holes. What is the...

Study smarter with the SolutionInn App


