For each of the following pairs of compounds, identify which compound would react more rapidly in an
Question:
(a)
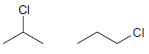
(b)
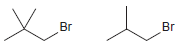
(c)
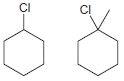
(d)

Transcribed Image Text:
CI .CI Вг Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a b c ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them: a. b. c. d. e. f. g. h. i. cis-2-butene and trans-2-butene j. CH3CH2CH2OH...
-
For each of the following pairs of compounds, indicate the compound that you would expect to be a more potent inhibitor of dihydrofolate reductase: a. b. NH NH CI of CH3 CH3 NH2 CH3 or H.NNCHCHCHCH,...
-
For each of the following pairs of compounds, determine which compound is more stable (you may find it helpful to draw out the chair conformations): (a) (b) (c) (d) II
-
In Exercises 1114, graph each equation in a rectangular coordinate system. If two functions are indicated, graph both in the same system. Then use your graphs to identify each relations domain and...
-
Identify three reasons why individuals create new business ventures. Next discuss any goals or motives that would drive you to become an entrepreneur. Provide examples for your response. Discuss at...
-
Compute parameter estimates for a regression model that is nonlinear in the parameters and explain how nonlinear least squares differs from linear least squares.
-
Use stepwise regression to build a model for the NBA Combine data in Table B.23. Perform a residual analysis on the final model. Compare this model to the all possible regressions model from Problem...
-
(Retained Earnings Statement, Prior Period Adjustment) Below is the retained earnings account for the year 2004 for LeClair Corp. (a) Prepare a corrected retained earnings statement. LeClair Corp....
-
What is the Present Value of receiving $100,000 in 20 years if interest rates are 5%? Reference the following formulas to solve the problem: FV=PV / (1+r)^t -1)/r PV=FV (1+r)^t PVA...
-
Assume that the Financial Management Corporations $1,000-par-value bond had a 5.700% coupon, matured on May 15, 2020, had a current price quote of 97.708, and had a yield to maturity (YTM) of 6.034%....
-
Draw all isomers of C 4 H 9 I, and then arrange them in order of increasing reactivity toward an S N 2 reaction.
-
Consider the following reaction: (a) How would the rate be affected if the concentration of the alkyl halide is doubled? (b) How would the rate be affected if the concentration of sodium cyanide is...
-
Match each equation with its graph. Explain your choices. (Dont use a computer or graphing calculator.) (a) y = x 2 (b) y = x 5 (c) y = x 8
-
How will total cost of ownership affect the way in which a business marketer prices its products?
-
Critically evaluate the methods that a bank might use to go about researching market potential for business development loans in a foreign country.
-
Compare and contrast the tasks performed by distributors and agents.
-
Parrish and Chlarson worked for J&K Computer Systems, Inc. Parrish was a computer programmer, and Chlarson was a trainee. In his capacity as programmer, Parrish wrote an accounts receivable program....
-
Describe the decision-making process enacted by organizational customers, identifying how and why this process might vary.
-
Suppose an investigator places first a free electron and then a free proton into an electric field between oppositely charged conducting plates. (a) How do the forces acting on the electron and those...
-
Making use of the tables of atomic masses, find the velocity with which the products of the reaction B10 (n, ) Li7 come apart; the reaction proceeds via interaction of very slow neutrons with...
-
Give structural formulas for the products that you would expect from the following reactions: (a) (b) (c) (d) KMnO, heat B-Pinene H2. Pt Zingiberene HCi Caryophyllene -Selinene (1) BH3 THF (2 equiv.)...
-
Draw the two basic ring systems given in Fig. 23.6 for the 5a and 5b series showing all hydrogen atoms of the cyclohexane rings. Label each hydrogen atom as to whether it is axial or equatorial.
-
(a) Androsterone, a secondary male sex hormone, has the systematic name 3a-hydroxy- 5a-androstan-17-one. Give a three-dimensional formula for androsterone. (b) Norethynodrel, a synthetic steroid that...
-
Jenny wishes to accumulate $10000 over 5 years for an overseas trip. She will make a deposit every 6-months, and the funds will earn the interest rate of 4% per annum compounded semi-annually. How...
-
Write a short C++ program that will prompt the user and input three integer values (month, day, year) from the console in a "date" format that includes '/' separator characters. e.g., 8/21/2013. Your...
-
Aimee is the owner of a stock with annual returns of 12.3 percent, -5.0 percent, 6.6 percent, and 18.2 percent for the past four years. She thinks the stock may achieve a return of 27 percent this...

Study smarter with the SolutionInn App


