For each of the following reactions, identify whether you would use hydroxide or tert-butoxide to accomplish the
Question:
a)

b)
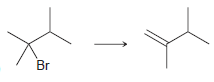
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a The more substituted alken...View the full answer

Answered By

Rehab Rahim
I am well versed in communicating and teaching in areas of all business subjects. I have helped many students in different ways from answering answers to writing their academic papers.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following reactions. provide the following information. (a) Give the structures of all products (including stereoisomers). (b) If more than one product is formed, give the...
-
For each of the following reactions, (1) indicate whether elimination will occur via an E2 or an E1 reaction, and (2) give the major elimination product of each reaction, ignoring stereoisomers: a....
-
For each of the following reactions, name the enzyme that catalyzes the reaction and name the required coenzyme: a. b. c. d. e. f. CH CSCoA ATP, Mg HCO3 OCCH2CSCOA 2+ (CH2)4C0- CH24CO SH SH S-S...
-
Perpetual inventory using FIFO Beginning inventory, purchases, and sales for Item Zeta9 are as follows: Oct. 1 Inventory 37 units @ $19 Oct. 7 Sale Oct. 15 Purchase Oct. 24 Sale Assuming a perpetual...
-
The mass of a sample of silty clay soil plus container is 18.43 g and the weight of the dry soil plus container is 13.67 g. The container weighs 8.84 g. Compute the water content of the sample.?
-
Use the minimum-variance method to estimate the PSD of \(L=256\) samples of the following signal: \[y(n)=\sin \frac{\pi}{2} n+x_{1}(n)\] where \[x_{1}(n)=-0.8 x_{1}(n-1)+x_{2}(n) \text {, }\] with...
-
Blockbuster, Inc., is the world's leading provider of rentable DVDs and videogames. Blockbuster estimates that 64 percent of the U.S. population lives within a 10-minute drive of a Blockbuster store....
-
Define each of the following terms: a. PV; i; INT; FVn; PVAn; FVAn; PMT; m; iNom b. FVIFi,n; PVIFi,n; FVIFAi,n; PVIFAi,n c. Opportunity cost rate d. Annuity; lump sum payment; cash flow; uneven cash...
-
Rousan contributed $1900.00 at the beginning of every month to an RRSP. Interest on the account is 4.95% compounded monthly. If she converted the balance after 13 years into an RRIF paying 7.1%...
-
2. An electronic component in an air-conditional system has a failure rate of 0.006 per minute. The expected time to failure for this component is 10000 hours. What proportion of this component would...
-
Describe pesticide resistance and discuss some methods for preventing the problems.
-
What are some of the long-term effects of pesticides on human health?
-
Assume variable manufacturing overhead is allocated using machine-hours. Give three possible reasons for a $30,000 favourable variable overhead efficiency variance.
-
ces Chuck Wagon Grills, Incorporated, makes a barbecue grill it sells for $210. Data for last year's operations follow: Units in beginning inventory Units produced Units sold Units in ending...
-
An aqueous solution of sodium hydroxide contains 20% NAOH by mass. It is desired to produce an 8.0% NaOH solution by diluting a stream of the 20% solution with a stream of pure water. Calculate the...
-
A reverse osmosis membrane unit concentrates a liquid food from 10 to 20% total solids by selectively removing water. If 100 lb/hr of feed is input to the membrane, how much concentrated product is...
-
8-23 A synthesis gas consists of 40 percent CO and 60 percent H, by volume. Determine it's higher and lower heating values. The higher and lower heating values of hydrogen are 141,800 kJ/kg and...
-
A chemist adds 280.0 mL of a 2.2 mol/L iron(II) bromide (FeBr2) solution to a reaction flask. Calculate the mass in kilograms of iron(II) bromide the chemist has added to the flask. Round your answer...
-
Air molecules stuck inside an inflated balloon collide continuously with the inner surface of the balloon. Each collision provides a little push outward on the balloon. All the many collisions...
-
Consider model (9.18). What is the effect on the model parameter estimates, their standard errors, and the goodness-of-fit statistics when (a) The times at risk are doubled, but the numbers of deaths...
-
What alkynes would you start with to prepare the followingketones? (a) (b) CHCHH2H CH3CH2CH2CH3
-
What alkyne would you start with to prepare each of the following compounds by a hydroboration/oxidationreaction? (b) (a) CH CH-CCHCH3 -CH2CH CH
-
How would you prepare the following carbonyl compounds starting from an alkyne (reddish brown ? Br)? (b) (a)
-
Sales quantity Sales Direct material Direct labor Manufacturing overhead Selling and Admin. Expenses The total cost of goods sold is 1,000 $66.00 per unit $12.00 $4.00 $3.00 per unit $1,800 per month...
-
Let R denote the region in the xy-plane that lies in the first quadrant and is bounded by the lines y = 1 - x, y = 2 - x, y = x + 1, and y = x - 1. Set u=x+y and v = y-x. (a) Solve for x and y in...
-
Reply to The culture established by Mr. Kelleher places importance on employee happiness, which directly influences customer satisfaction and overall organizational achievements. To maintain this...

Study smarter with the SolutionInn App


