Question: For each pair of compounds below, predict which compound will have the higher boiling point and explain your choice: a) CH 3 CH 2 CH
For each pair of compounds below, predict which compound will have the higher boiling point and explain your choice:
a) CH3CH2CH2OCH3 or CH3CH2CH2CH2OH
b) CH3CH2CH2CH3 or CH3CH2CH2CH2CH3
c)
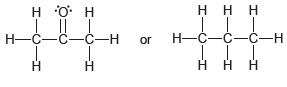
or
Step by Step Solution
3.40 Rating (163 Votes )
There are 3 Steps involved in it
a The second because it po... View full answer

Get step-by-step solutions from verified subject matter experts


