6. Which of the compound would have a higher boiling point: (a) cyclohexene or (b) 1,2-...
Fantastic news! We've Found the answer you've been seeking!
Question:
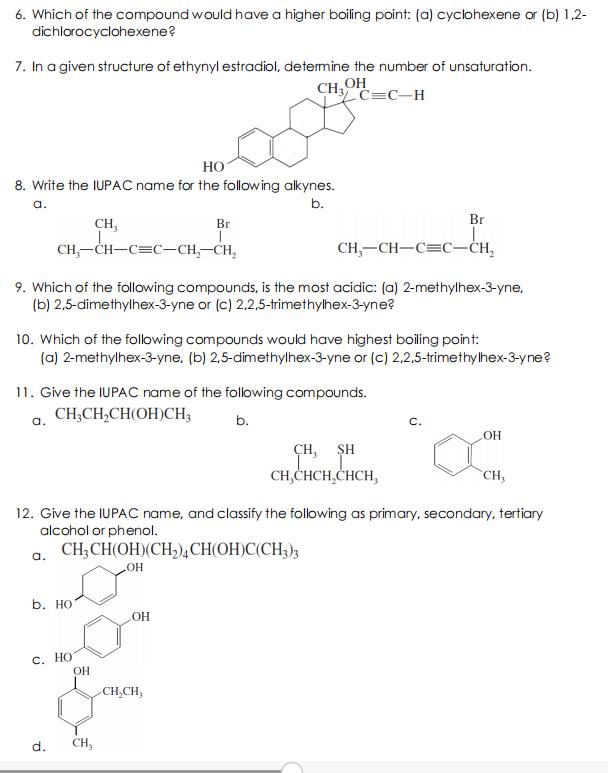
Transcribed Image Text:
6. Which of the compound would have a higher boiling point: (a) cyclohexene or (b) 1,2- dichlorocyclohexene? 7. In a given structure of ethynyl estradiol, determine the number of unsaturation. CH OH C=C-H HO 8. Write the IUPAC name for the following alkynes. a. b. Br CH, Br CH,-CH-C=C-CH,-CH, CH,-CH-C=C-CH, 9. Which of the following compounds, is the most acidic: (a) 2-methylhex-3-yne, (b) 2,5-dimethylhex-3-yne or (c) 2,2,5-trimethylhex-3-yne? 10. Which of the following compounds would have highest boiling point: (a) 2-methylhex-3-yne. (b) 2,5-dimethylhex-3-yne or (c) 2,2,5-trimethylhex-3-yne? 11. Give the IUPAC name of the following compounds. CH,CH,CH(OH)CH; a. b. C. CH, SH CH,CHCH,CHCH, CH 12. Give the IUPAC name, and classify the following as primary, secondary, tertiary alcohol or phenol. a. CH;CH(OH)(CH,),CH(OH)C(CH3)3 b. НО ОН С. Но CH,CH, d. CH, 6. Which of the compound would have a higher boiling point: (a) cyclohexene or (b) 1,2- dichlorocyclohexene? 7. In a given structure of ethynyl estradiol, determine the number of unsaturation. CH OH C=C-H HO 8. Write the IUPAC name for the following alkynes. a. b. Br CH, Br CH,-CH-C=C-CH,-CH, CH,-CH-C=C-CH, 9. Which of the following compounds, is the most acidic: (a) 2-methylhex-3-yne, (b) 2,5-dimethylhex-3-yne or (c) 2,2,5-trimethylhex-3-yne? 10. Which of the following compounds would have highest boiling point: (a) 2-methylhex-3-yne. (b) 2,5-dimethylhex-3-yne or (c) 2,2,5-trimethylhex-3-yne? 11. Give the IUPAC name of the following compounds. CH,CH,CH(OH)CH; a. b. C. CH, SH CH,CHCH,CHCH, CH 12. Give the IUPAC name, and classify the following as primary, secondary, tertiary alcohol or phenol. a. CH;CH(OH)(CH,),CH(OH)C(CH3)3 b. НО ОН С. Но CH,CH, d. CH,
Expert Answer:
Answer rating: 100% (QA)
6 12dichlorocyclohexene has higher boiling point due to dipole moment present in the molecule Also d... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
Which of the following would have a higher standard deviation? (a) IQ of students on your campus (b) IQ of residents in your home town? Why?
-
Write the IUPAC name for each of the following coordination compounds. a. K3[FeF6] b. [Cu(NH3)2(H2O)2]2+ c. (NH4)2[Fe(H2O)F5] d. [Ag(CN)2]
-
Explain why a. H2O has a higher boiling point than CH3OH(65oC). b. H2O has a higher boiling point than NH3(- 33oC). c. H2O has a higher boiling point than HF (20C).
-
Partners have fiduciary obligations to each other. Explain what this means and give examples.
-
The beginning inventory for Tschabold Co. and data on purchases and sales for a two-month period are shown in Problem 6-3A. Round the average cost to two decimal places. In Problem 6-3 A The...
-
What are the two criteria for determining an SME?
-
Your firm uses a large parallel-plate capacitor to store energy, and you measure the electric field strength between the plates to determine the amount of energy stored. During a test run with a new...
-
Hamilton and Battles, Ltd. produces and sells two products-guitar cases and violin cases. Each of these products is made in a dedicated manufacturing facility, and the product line managers are...
-
Men Conference Ltd has an authorized share capital of 60,000,000 ordinary shares of Sh. 10 each and 500,000 shares of Sh. 50 each. The following trial balance of the company was extracted as at 31.12...
-
1. What problems might have contributed to the firms poor performance? 2. Although several problems were encountered in implementing the business plan, the primary reason for low profits turned out...
-
8. (12 Points) Convert the following assembly language program to C/C++. Note that you do NOT need to specify in the C program the code memory locations for the MAIN (0x100) subroutine. .ORG 0 LDI...
-
Jack works for Peterson Millworks, is married, and has two children. Peterson Millworks offers a PPO to its 50 employees. If Jack and his wife Alice were to divorce, which of the following is or are...
-
As computer structures get smaller and smaller, quantum rules start to create difficulties. Suppose electrons move through a channel in a microprocessor. If we know that an electron is somewhere...
-
Jay believes the Commerce Clause enhances the governments power to regulate business, but Ellie believes the Commerce Clause restricts the governments power in this regard. How could both be correct...
-
Which of the following is not an essential health benefit under the Affordable Care Act (ACA)? A. Ambulatory patient services. B. Laboratory services. C. Maternity and newborn care. D. Vision care.
-
Assets held in retirement plans reported on the FAFSA. Elective deferrals reported on the FAFSA and are counted as untaxed income. A. Are, are not. B. Are, are. C. Are not, are. D. Are not, are not.
-
At May 3 1 , 2 0 2 2 , the accounts of Lopez Company show the following. May 1 inventories - finished goods $ 1 2 , 6 0 0 , work in process $ 1 4 , 7 0 0 and raw materials $ 8 , 2 0 0 . May 3 1...
-
Organizations are increasing their use of personality tests to screen job applicants. What are some of the advantages and disadvantages of this approach? What can managers do to avoid some of the...
-
Use the numbers for the alcohol and tobacco category from the table in the Application "Goods with a Large Consumer Surplus Loss from Price Increases" to draw a figure that illustrates the roles that...
-
Change the answer given in the Challenge Solution for the short run rather than for the long run.
-
Because many consumers choose between coffee and tea, the coffee and tea demand functions depend on both prices. Suppose the demand curves for coffee and tea are where \(Q_{c}\) is the quantity of...

Study smarter with the SolutionInn App

