For each pair of the following compounds identify which compound would react more rapidly in an E2
Question:
a.
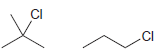
b.
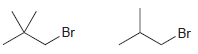
Transcribed Image Text:
CI .CI Br Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a The first compound will react more rapidly ...View the full answer

Answered By

Atuga Nichasius
I am a Highly skilled Online Tutor has a Bachelor’s Degree in Engineering as well as seven years of experience tutoring students in high school, bachelors and post graduate levels. I have a solid understanding of all learning styles as well as using asynchronous online platforms for tutoring needs. I individualise tutoring for students according to content tutoring needs assessments.
My strengths include good understanding of all teaching methods and learning styles and I am able to convey material to students in an easy to understand manner. I can also assists students with homework questions and test preparation strategies and I am able to help students in math, gre, business , and statistics
I consider myself to have excellent interpersonal and assessment skills with strong teaching presentation verbal and written communication
I love tutoring. I love doing it. I find it intrinsically satisfying to see the light come on in a student's eyes.
My first math lesson that I taught was when I was 5. My neighbor, still in diapers, kept skipping 4 when counting from 1 to 10. I worked with him until he could get all 10 numbers in a row, and match them up with his fingers.
My students drastically improve under my tutelage, generally seeing a two grade level improvement (F to C, C to A, for example), and all of them get a much clearer understanding!
I am committed to helping my students get the top grades no matter the cost. I will take extra hours with you, repeat myself a thousand times if I have to and guide you to the best of my ability until you understand the concept that I'm teaching you.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which isomer reacts more rapidly in an E2 reaction, cis-1-bromo-4-tert-butylcyclohexane or trans-1-bromo-4-tert-butylcyclohexane? Explain your choice.
-
For each pair of the following molecules, indicate whether its members are identical, structural isomers, conformers, or stereoisomers. How would you describe the relation between conformations when...
-
For each pair of compounds below, identify which compound is more acidic and explain your choice: (a)
-
A restaurant records the following data over a month for its food: Opening inventory: $31,000 Purchases: $88,000 Closing Inventory: $28,000 Transfers in: $800 Transfers out: $200 Employee meals:...
-
1. Toward which of the three main ethical approaches (duty based, consequence based, virtue based) do you most lean? To what extent are you egoist in your approach to life? 2. What do you see as the...
-
Suppose that events A 1 , A 2 , and A 3 form a partition of the sample space S with P(A 1 ) = 0.35, P(A 2 ) = 0.45, and P(A 3 ) = 0.2. If E is an event that is a subset of S and P(E|A 1 ) = 0.25,...
-
How do we discover business activities?
-
On January 1, 2014, Jade Company issued $2,000,000 face value, 7%, 10-year bonds at $2,147,202. This price resulted in a 6% effective-interest rate on the bonds. Jade uses the effective-interest...
-
Alexa owns a condominium near Cocoa Beach in Florida. In 2023, she incurs the following expenses in connection with her condo: Insurance Mortgage interest $ 2,500 Property taxes Repairs & maintenance...
-
In a cellular system, diffraction, reflection, and direct path take different amount of time for the signal to reach an MS. How do you differentiate and use these signals? Explain clearly. Compute...
-
1-Bromobicyclo [2.2.2] octane does not undergo an E2 reaction when treated with a strong base. Explain why not.
-
A purely competitive firm finds that the market price for its product is $20. It has a fixed cost of $100 and a variable cost of $10 per unit for the first 50 units and then $25 per unit for all...
-
Would the constitutional right of privacy provide a defense to a charge of possession of obscene materials where videotapes were viewed only by the defendant in the privacy of his or her home?
-
A uniform bridge 30 m long and weighing 6*10^(3) N is supported by two pillars located 5 m from each end. A 4*10^(8) N car is parked 6 m from the left end of the bridge. Calculate the force the...
-
The doctor writes an order IV bolus. The order said to administer to IV now 12.5 mg you are supplied with a via That is labeled 50m/ml.how many ml will you draw up and administer per dose ?
-
Let f be the function f(x)= x^2 ln(x). For what value of x is the slope of the line tangent to the graph of f at (x,f(x)) equal to 2.
-
solve for t 1 2 0 0 = 1 0 ( 2 t + 2 ) ^ 2 - 2 0 ( 2 t + 2 ) + 5 0 0
-
Solve the following integral and show your work. d.) gu)=sin(3x) xg(x)dx
-
Fill in the blank with an appropriate word, phrase, or symbol(s). A distinct group of objects without regard to their arrangement is called a(n) _____________ .
-
How many years will it take a $700 balance to grow into $900 in an account earning 5%?
-
Would you expect two diastereomers such as meso-2, 3-dibromobutanc and (2R, 3R)-dibromo butane to have identical or different IR spectra? Explain.
-
Propose structures for compounds that meet the following descriptions: (a) C5H8, with IR absorptions at 3300 and 2150 cm1 (b) C4H80, with a strong IR absorption at 3400 cm1 (c) C4H80, with a strong...
-
How could you use infrared spectroscopy to distinguish between the following pairs of isomers? (a) HC CCH2NH2 and CH3CH2C N (b) CH3COCH3 and CH3CH2CHO
-
In February 2 0 1 6 , hackers targeted the central bank of Bangladesh and exploited vulnerabilities in SWIFT, the global financial system s main electronic payment messaging system, trying to steal...
-
In the Heckscher-Ohlin model, as trade occurs in a capital-abundant country, increased imports will force domestic import- competing firms to decrease prices and production. Labor and capital will...
-
Why has Gatorade been so successful in the sports drink market?

Study smarter with the SolutionInn App


