How many constitutional isomers are obtained when each of the following compounds undergoes monochlorination? (a) (b) (c)
Question:
(a)
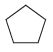
(b)
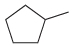
(c)
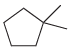
(d)
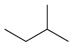
(e)
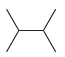
(f)
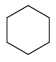
(g)
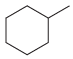
(h)
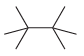
(i)
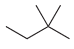
(j)

Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a 1 b 4 ...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds: a. b. c. d. e. f. g. h. i. COOH Br Br Br Br CH3 ,. CHCH NO. OCH, CH2CH3 SOH CI CI CH3 CH2CH3 Cl
-
Name the following compounds: a. b. c. d. e. f. g. h. i. j. CH2CH3 CH,CH-CHCH-CH-CH-COH CH.CH,COCH.CH.CH. CH CH,CH CH C N CH.CH COCCH.CH CH3CH2COCCH2CH CH3CH2CH2CN(CH3)2 CH3CH2CH CH2CC COCCH CH CHCH...
-
Give appropriate names for the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) CH2 Ph-CO-CH CH CH NH C-Ph Ph-OC CH Ph-C-O-CH CN CH30-C CH3 C-N(CH CH32 CH;CH2
-
On the cost of goods manufactured schedule, depreciation onfactory equipment... A. is not listed because it is not a product cost. B. is not an inventoriable cost. C. is not listed because it is...
-
The current operation consists of the following elements: Operator removes pressed unit (0.2 min) Operator walks to inspection area, checks for defects (0.1 min) Operator files rough edges (0.2 min)...
-
Go to the Fidelity, CNN, Edward Jones, SmartMoney, AARP, and so on, retirement worksheets. Put in financial information that applies to someone between 40 and 60 such as a parent, aunt/uncle, and so...
-
Write requirements for the following products and services: a. Computer. b. Airplane. c. Food Processor. d. Online Banking e. Life Insurance. f. Party Planning.
-
Estimate Walmarts weighted average cost of capital (WACC). State any assumptions and your methodology in a clear and concise manner. EXHIBIT 2: WALMART INC., CONSOLIDATED BALANCE SHEET AS OF JANUARY...
-
Orders have been coming in like crazy since the special order bundle sale started! Aaliyah has asked Missy to help them out with packing and shipping everything. Together, they were able to get the...
-
Elsas financial year ends on 31 March. She depreciates her office equipment at 20% per annum on cost. Depreciation is calculated from the date of purchase. On 1 April the balances in Elsas books...
-
What are the null and research hypotheses for correlations?
-
Briefly describe what you can learn about a target protein by using each of these two techniques.
-
A phone service center keeps track of the number of customers it services for a random 1-hour period during the day. The distribution is shown. Find the mean, variance, and standard deviation for the...
-
A Major Disciplinary Infraction for Possession of a Weapon in school is a violation that occurs in school and severely interferes with the learning and safety of others, and is of a threatening or...
-
The court found a blanket license agreement to not present a per se violation of the Sherman act, with respect to transaction costs. what are some of the transaction costs involved in buying the...
-
If women can participate in men's teams, should men be allowed to compete on women's teams? Discuss.
-
Describe and discuss police discretion, and the various control mechanisms available i.e., internal control mechanisms, external control mechanisms, control by the citizens, legislative control, and...
-
Explain in detail the concept of Global Dexterity - what does it mean. Provide an example of how it can be used effectively in a business context.
-
Frey Company issued bonds of $300,000 face value on January 1, Year 1. The bonds had a 6 percent stated rate of interest and a 10-year term. Interest is paid in cash annually, beginning December 31,...
-
What mass of KBr (in grams) should you use to make 350.0 mL of a 1.30 M KBr solution?
-
While working in the chemical stockroom, you discover an unlabeled bottle containing a liquid compound. You carefully smell the liquid and discover that it has a fishy odor. What functional group do...
-
Turpentine, obtained from pine trees, is composed primarily of ?-pinene and ?-pinene, Explain whether you expect turpentine to mix with water. If a point dissolves in turpentine, what does this...
-
The structure of a typical fat is shown here. Estimate the energy content of fat compared to the other compounds discussed in the Focus On box on p. 146 and explain yourreasoning. CH OC(CH) 16CH3 O...
-
Jenny wishes to accumulate $10000 over 5 years for an overseas trip. She will make a deposit every 6-months, and the funds will earn the interest rate of 4% per annum compounded semi-annually. How...
-
Write a short C++ program that will prompt the user and input three integer values (month, day, year) from the console in a "date" format that includes '/' separator characters. e.g., 8/21/2013. Your...
-
Aimee is the owner of a stock with annual returns of 12.3 percent, -5.0 percent, 6.6 percent, and 18.2 percent for the past four years. She thinks the stock may achieve a return of 27 percent this...

Study smarter with the SolutionInn App


