How many signals do you expect in the 1 H NMR spectrum of each of the following
Question:
(a)
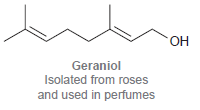
(b)
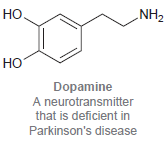
(c)
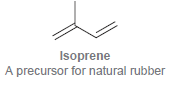
Transcribed Image Text:
ОН Geraniol Isolated from roses and used in perfumes но. NH2 но Dopamine A neurotransmitter that is deficient in Parkinson's disease
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a Ni...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many signals do you expect in the 13 C NMR spectrum of each of the following compounds? a. b. c. d. Br
-
Consider the following compound: (a) How many signals do you expect in the 1 H NMR spectrum of this compound? (b) Rank the protons in terms of increasing chemical shift. (c) How many signals do you...
-
Dimethylformamide (DMF) is a common solvent: (a) The 1 H NMR spectrum of DMF exhibits three signals. Upon treatment with excess LAH followed by water, DMF is converted into a new compound that...
-
discuss case study a bout remote analysis during covid 1 9 - 1 9 virus
-
What are two examples of industries that could benefit from IoM?
-
SLIX wax is developing a new high performance fluorocarbon wax for cross country ski racing designed to be used under a wide variety of conditions. In order to justify the price marketing wants, the...
-
Every day, logistics companies such as United Parcel Service (UPS) must decide how to route their trucks, that is, the order in which to deliver the packages that have been loaded on a truck. UPS...
-
Juras Inc. and Hinson Inc. have the following operating data: a. Compute the operating leverage for Juras Inc. and Hinson Inc. b. How much would income from operations increase for each company if...
-
Kingsport Containers Company makes a single product with wide seasonal variations in demand. The company uses a job-order costing system and computes plantwide predetermined overhead rates on a...
-
The number of surface flaws in plastic panels used in the interior of automobiles has a Poisson distribution with a mean of 0.05 flaw per square foot of plastic panel. Assume an automobile interior...
-
Daniel Barnes, financial manager of New York Fuels (NYF), a heating oil distributor, is concerned about the companys working capital policy, and he is considering three alternative policies: (1) a...
-
Rank the signals of the following compound in terms of increasing chemical shift. Identify the proton(s) giving rise to each signal: CI. .
-
In 150 words or fewer, explain the difference between horizontal and vertical analysis. Be sure to include in your answer how each might be used.
-
Analyze the events surrounding the sale of FHs land and buildings. Is it a subsequent event? If so, which type? Fabrication Holdings Ltd. (FH) has been a client of KFP Partners for many years. You...
-
Using a semiannually compounded yield of \(4 \%\) p.a., numerically or via evaluating the formula, compute the PV01 of (a) \(\$ 1\) million 5 -year zero-coupon bond (b) \(\$ 1\) million 5 -year \(4...
-
In 2016, Susan Christian transferred a machine with a fair market value of \(\$ 30,000\) and an adjusted basis of \(\$ 40,000\) to JPC Corporation in return for 60 shares of stock. The stock...
-
Within a clients IT system, supplier information is contained in a supplier master file (SMF). Each supplier has a unique supplier code. If the purchasing clerk attempts to place an order from a...
-
Using the bisection method, compute the semiannual yield of a 5-year bond paying a semiannual coupon of \(2 \%\) per annum and trading at \(98 \%\). Start with bracketing levels of \(0 \%, 10 \%\),...
-
Susan and Stan Collins live in Iowa, are married and have two children ages 6 and 10. In 2018, Susans income is $38,290 and Stans is $12,000 and both are self-employed. They also have $500 in...
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
In humans, the final product of purine degradation from DNA is uric acid, pKa = 5.61, which is excreted in the urine. What is the percent dissociation of uric acid in urine at a typical pH = 6.0? Why...
-
Shown here are some pKa data for simple dibasic acids. How can you account for the fact that the difference between the first and second ionization constants decreases with increasing distance...
-
Predict the product of the reaction of p-methyl benzoic acid with each of the following: (a) LiAlH 4 , then H 3 O + (b) N-Bromosuccinimide in CCl 4 (c) CH 3 MgBr in ether, then H 3 O + (d) KMnO 4 , H...
-
Salmon ASA has just issued a callable seven-year, 8% coupon bond with coupon payable annually. The bond can be called at par in two years or anytime thereafter on a coupon payment date. It has a...
-
Lamda corporation wants to acquire another company within its industry for $100m and it expects the acquisition to contribute to its free cash flow by $5m the first year, and this contribution is...
-
Dewan INC. has several divisions, each with a manager responsible for the operations of the division. Each division of Dewan controls product design, sales, pricing, operating costs, and profits.....

Study smarter with the SolutionInn App


