How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29
Question:
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy?
Problem 15.29
a.
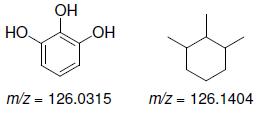
b.
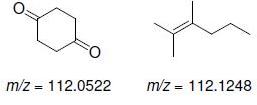
Transcribed Image Text:
OH HO. HO m/z = 126.0315 m/z = 126.1404
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a The first compound should have a very broad signal between 3200 and 3600 cm 1 co...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you distinguish between each pair of compounds using mass spectrometry? a. b. N.
-
How would you distinguish between each pair of compounds using high-resolution mass spectrometry? a. b. .
-
How would you distinguish between the following pairs of compounds using simple chemical tests? Tell what you would do and what you would see. (a) Cyclopentane and cyclopentane (b) 2-Hexene and...
-
If $4000 is deposited into an account paying 3% interest compounded annually and at the same time $2000 is deposited into an account paying 5% interest compounded annually, after how long will the...
-
Here is the article from 'Required Resources': abcnews.go.com/Health/story?id=117304&page=1 Read this article on the dangers of roller coasters. After you've read the article, write a short summary...
-
Use Eq. (17.1) to find the minima of Fig. 17.2 . Confirm Eq. (17.5) for \(\mu^{2})^{1 / 2}=\) \(\left(-2 \mu^{2} ight)^{1 / 2}\) after the symmetry is broken spontaneously. Data from Eq. 17.1 Data...
-
Prove that \(B(x ; n, p)=1-B(n-x-1 ; n, 1-p)\).
-
Rodamex Inc. reported the following summary transactions during the year ended December 31, 2015: a. Sold merchandise for $ 5,000,000, including $ 500,000 cash and $ 4,500,000 on account, with terms...
-
Direct Materials Used, Cost of Goods Manufactured In September, Lauren Ashley Company purchased materials costing $210,000 and incurred direct labor cost of $140,000. Overhead totaled $320,000 for...
-
What is the solution of the recursive equation T (n) = 0.015625T +n?? (1/4) a) O(n') b) O(n' lg n) c) O(n* Ign) d) O(7*)
-
Rank each of the bonds identified in order of increasing wave number. R-CEN - N-H - -
-
How many signals do you expect in the 13 C NMR spectrum of each of the following compounds? a. b. c. d. Br
-
The first and second terms of a geometric sequence are a and ab, in that order. What is the 643rd term of the sequence? A. (ab) 642 B. (ab) 643 C. a 642 b D. a 643 b E. ab 642
-
Of the ground rules listed in critical concept 7.1, which three are most important for a team to adopt, and why? When choosing the rules, consider your past experiences working with a team or a...
-
Why are two control charts not necessary in controlling for attributes? Might not the variability of the fraction defective or the number of defects also be going out of control?
-
Contrast the cost-schedule reconciliation charts with the earned value chart. Which one would a PM prefer?
-
How might the idea of total quality control be applied to a distributed organisation such as a supply chain?
-
List six specific quality control responsibilities of audit engagement partners.
-
After a fuel rod in a fission reactor reaches the end of its life cycle (typically 3 years), most of the energy that it produces comes from the fissioning of plutonium-239. How can this be?
-
Define deferred revenue. Why is it a liability?
-
Iodine azide,I-Nr, adds to isobutylene in the following manner: iodine azide
-
For each of the following cases, provide the structure of an alkene that would give the alcohol as the major (or only) product of hydroboration--oxidation. CH CH CH CH OH CH, CH,
-
Which of the following alkenes would yield the same alcohol from either oxymercuration-reduction or hydroboration--oxidation, and which would give different alcohols? Explain. (a) cis-2-butene (b)...
-
Suppose you were interested in studying the quality of conditions within a prison. What indicators would you measure to give the clearest picture of the realities of prison life? Cite the below...
-
On March 31, 2023, Panda Co. assessed its assets for impairment as part of its year-end procedures. It was found that equipment had a recoverable value of $15,000, a remaining useful life of three...
-
Petty's comparative balance sheets at December 31, 2020, and December 31, 2019, report the following (in millions). (Click the icon to view the comparative balance sheets.) Requirements Below are...

Study smarter with the SolutionInn App


