Rank each of the bonds identified in order of increasing wave number. R-CEN - N-H - -
Question:
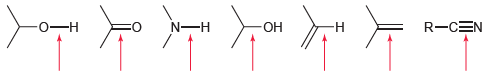
Transcribed Image Text:
R-CEN -о—н N-H -ОН -н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
OH NH In...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank each of the following series of compounds in order of increasing oxidationlevel: CI (a) (b) CH3CN CH3CH2NH2 H2NCH2CH2NH2
-
Rank the following bonds in order of increasing ionic character: NOO, CaOO, COF, BrOBr, KOF.
-
The partial Lewis structure that follows is for a hydrocarbon molecule. In the full Lewis structure, each carbon atom satisfies the octet rule, and there are no unshared electron pairs in the...
-
Write the expression as one ratio without any negative exponents. x1/4x-3/4 X
-
Discuss the growing income inequality in the U.S. and the world with someone mature enough to have some perspective from experience about it. Ask them what they think about the widening gap between...
-
Jeff has \(\$ 10,000\) to invest for a period of 5 years. The following account is available at his bank. The account pays 4 percent/year for year one, 6 percent/year for year two, 8 percent/year for...
-
Prove that \(b(x ; n, p)=b(n-x ; n, 1-p)\).
-
Following are descriptions of transactions and other financial events for the City of Tetris for the year ending December 2010. Not all transactions have been included here. Only the General Fund...
-
As of December 31 of the current year, Armani Company's records show the following. Cash Accounts receivable $ 11,800 10,800 Supplies 7,800 Equipment 6,800 Accounts payable 14,600 Common stock 15,800...
-
Read the following two cases and explain why the Supreme Court reached different conclusions for cases involving similar facts and issues: CIR v. Court Holding Co., 33 AFTR 593, 45-1 USTC 9215...
-
All of the following compounds absorb IR radiation in the range between 1600 and 1850 cm -1 . In each case, identify the specific bond(s) responsible for the absorption(s), and predict the...
-
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29 a. b. OH HO. HO m/z = 126.0315 m/z = 126.1404
-
What evidence do you see of a commitment to social responsibility at Chevron? If Chevron were to leave Venezuela, what do you think would be the impact on its employees and the communities where it...
-
You have the following variable defined: Based on attack, we want to figure out what type the attack is weak against and strong against. Create a series of if/else-if/else statements to print the...
-
Consider how you might pursue mistake-proofing in a digital or digitally enabled organisation?
-
Explain briefly three motivations for companies to engage in corporate responsibility reporting.
-
Practice evaluating the five forces, using the strategy tools presented in this chapter. 1. Use either an industry that your professor assigns to you or pick an industry of your choice. 2. Use a...
-
In your opinion, does it make more sense for an organization to merge its lean and Six Sigma programs or keep them separate?
-
Why is a nuclear fusion reaction so difficult to induce?
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
The industrial synthesis of methyl tert-butyl ether involves treatment of 2-methylpropene with methanol (CH3OH) in the presence of an acid catalyst, as shown in the following equation. CH3 H3C H3C...
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
After you've completed this week's assigned reading, watch the video below. Next, respond to the Discussion questions underneath. Your first post should answer those questions and should be made by...
-
Factorize (x4 - 20x + 100)
-
Paragraph discussing food insecurity in the United States. What agencies in the federal government play an important role in ameliorating this situation? What groups of people are at particular risk?

Study smarter with the SolutionInn App


