Question: Hydroxide is not a suitable base for deprotonating acetylene: Explain why not. Can you propose a base that would be suitable? - -: : -
Hydroxide is not a suitable base for deprotonating acetylene:
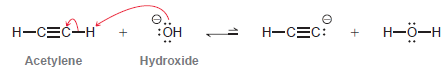
Explain why not. Can you propose a base that would be suitable?
- -: : - Acetylene Hydroxide
Step by Step Solution
3.48 Rating (161 Votes )
There are 3 Steps involved in it
The equilibrium does not favor deprotonation of acetylene by hydroxi... View full answer

Get step-by-step solutions from verified subject matter experts


