Identify the product(s) in each of the following reactions: (a) (b) (c) (d) Br ETOH
Question:
Identify the product(s) in each of the following reactions:
(a)
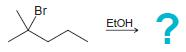
(b)
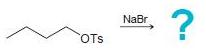
(c)
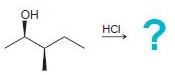
(d)
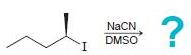
Transcribed Image Text:
Br ETOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a b...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the product of each of the following reactions: a. b. c. d. CH CH3 A CH2CH3 CH2CH 2CH3 -A CH2CH3 h CH2CH3 CH2CH3 CH2CHs
-
Predict the product(s) in each of the following aldol condensations. acetophenone + hexanal + hexanal NH
-
Identify the alkene product in each of the following Wittig reactions: (a) (b) ButanaH5)2P CHCH CH2 Cyclohexyl methyl ketone(H)2P CH2
-
Which statement is correct? A) Tax credits reduce tax liability on a dollar-for-dollarbasis. B) Tax deductions reduce tax liability on a dollar-for-dollarbasis. C) The benefit of a tax credit depends...
-
What are the key concerns in the design of a power tool?
-
What is Stats?
-
What are the advantages of using the Internet to conduct preliminary job interviews? What problems is a company that uses computer-aided interviewing likely to encounter?
-
As part of the audit of different audit areas, auditors should be alert for the possibility of unrecorded liabilities. For each of the following audit areas or accounts, describe a liability that can...
-
(13%) Problem 4: A student standing on a cliff throws a stone from a vertical height of d = 8.0 m above the level ground with velocity vo = 18 m/s at an angle 0 = 32 below the horizontal, as shown....
-
Based on Problem 2-9B, prepare an income statement for David Segal for the month of October 20. Problem 2-9B: David Segal started a business. During the first month (October 20--), the following...
-
Draw a relative energy diagram showing the conformational analysis of 1,2-dichloroethane. Clearly label all staggered conformations and all eclipsed conformations with the corresponding Newman...
-
Assign IUPAC names for each of the following compounds: (a) (b) (c) (d) . CH CH3
-
Valis Corporation had the following tax information. In 2008 Valis suffered a net operating loss of $450,000, which it elected to carry back. The 2008 enacted tax rate is 29%. Prepare Valiss entry to...
-
At the transport layer , to define the processes we need two identifiers _ _ _ _ _ _ _ _ _ _ _ _ .
-
where should you perform and display the results of error checking?
-
Calculate the goodwill in the following: Goodwill is to be calculated at one year's purchase of the last 3 years' average profit. The profit of the first year was Rs. 6,000, second year twice the...
-
Performance reviews are an integral part of which component of internal control? Multiple Choice Risk assessment. Control environment. Monitoring. Control activities
-
The capital employed in a company is Rs. 5,00,000. The normal rate of return is 20%. During the last four years the actual returns wewre : 24 %, 26%, 28%, and 30% on capital. The goodwill of the...
-
Albright was hired to be a bodyguard for Madonna. He became romantically involved with her for two years. Several years later he contracted to sell information about Madonna for a biography. One...
-
Consider the setup in Problem 16. Show that the relative speed of the ball and the point of contact on the stick is the same before and immediately after the collision. (This result is analogous to...
-
Benzene and hexane are both liquids at room temperature. Do you expect benzene and hexane to be miscible? Do you expect benzene and water to be miscible? Explain. Hexane 0 Benzene
-
One of these isomers is miscible with water, and the other is nearly insoluble. Explain. CHCHCHCOH CH,COCHCH3
-
Because of two hydrogen bonds, carboxylic acids show a very strong attractive force between two molecules that persists even in the gas phase. Show this hydrogen bonding between two carboxylic acid...
-
The quality-control department of Starr Communications, the manufacturer of video-game DVDs, has determined from records that 1.6% of the DVDS sold have video defects, 0.9% have audio defects, and...
-
What is the half-life in hours of procainamide in a patient whose total clearance is estimated to be 20 L/h and the volume of distribution is 120 L assuming one compartment, open model, first- order...
-
1. The technologies, such as ChatGPT, has been launched before introducing even a baseline of safety measures. This goes back to trade-offs that companies make, and people just watch the impact from...

Study smarter with the SolutionInn App


