Assign IUPAC names for each of the following compounds: (a) (b) (c) (d) . CH
Question:
(a)
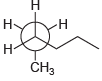
(b)
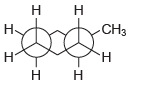
(c)
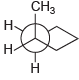
(d)
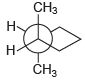
Transcribed Image Text:
Н Н н н. CH н н На CH3 Н н н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a Hexane b Meth...View the full answer

Answered By

Moses mwangi
With prior writing experience, be sure that I will give a great grade, If not an A+, it will be something close to this. My reviews speaks it all, Try me!!
4.80+
78+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Assign an IUPAC name for each of the following compounds. a. b. c. d. e. f. g. SH
-
Assign an IUPAC name for each of the following compounds: a. b. c. d. ,
-
Give the IUPAC names for each of the following compounds: (a) CH2CH(CH2)5CHCH2 (b) (c) (CH2CH)3CH (d) (e) (f) CH2CCHCHCHCH3 (g) (h) CH3 CHz CH3 HH CI CI H H H3C CH,CH2 CH2CH3
-
Table # 11.1.1: Autism Versus Breastfeeding Breast Feeding Timelines Autism Yes No None 241 20 Column Total 261 Less than 2 months 198 25 223 2 to 6 months 164 27 191 More than 6 months 215 44 259...
-
Design an ergonomic handle indicating all of the principles used in the design.
-
Let \(\left(\mathcal{C}_{(\mathrm{o})}, \mathscr{B}\left(\mathcal{C}_{(\mathrm{o})} ight), \mu ight)\) be the canonical Wiener space (we assume \(d=1\) ). We will now consider the space...
-
Dave Parrett, sales manager for Ace Chemicals, is wrestling with the issue of how to get Kay Powers back on track. Kay has been with the company for 20 years. Historically, she had been one of the...
-
Lanza Research Inc. manufactures high-quality hair care products in California. Copyrighted labels are attached to all products and packaging. In the United States, Lanza sells exclusively to...
-
Table 6. Steel Bolt Specific Heat Data Bolt T, (C) Water T (C) Water and Bolt T, (C) Water AT = T- T Bolt Bolt AT (C) Water Mass Mass (kg) (kg) Steel Bolt 85 18 20 2C 65 0.0015 0.15 Post-Lab...
-
Understand the process of mobile manufacturing company commencing from raw material decision to final delivery to the end consumer. Make a flow chart and suggest a strategy for procurement, vendor...
-
Identify the product(s) in each of the following reactions: (a) (b) (c) (d) Br ETOH
-
Some plants grow in soils as high as 20% iron. If the iron is present in the form of Fe(OH)3, the plants can still be iron deficient. Explain how this can be true. How can the iron in the soil be...
-
Use the graph to determine if the sequence is arithmetic or geometric. If the sequence is arithmetic, state the sign of the common difference d and estimate its value. If the sequence is geometric,...
-
Which Step completed to create a custom entity with text field and calculated datetime field?
-
2. Write code for a SmartPhone class which is an InternetThing that includes: a. A constructor which accepts a manufacturer(string), a model(String), a serial number(String) and the number of...
-
A pipe fitter must connect a pipeline to a tank as shown in the figure. The run from the pipeline to the tank is 62 ft 6 in., while the set (rise) is 38 ft 9 in. (a) How long is the connection?...
-
The last six times you purchased a stock you earned high returns within one year. Thus, you believe you will have the same result with your next stock purchase. This is an example of which one of the...
-
Isabella Flowers Unlimited held callable 8% bonds with a face amount of $59 million that were issued for $59 million on June 30, 2018. The bonds mature on June 30, 2023. Bondholders have the option...
-
Robert B. represented to Kathleen K. that he was free from venereal disease when he knew or should have known he had herpes. Relying on this representation, Kathleen K. had sexual intercourse with...
-
Three successive resonance frequencies in an organ pipe are 1310, 1834, and 2358 Hz. (a) Is the pipe closed at one end or open at both ends? (b) What is the fundamental frequency? (c) What is the...
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
One of the isomers of C 5 H 12 reacts with Cl 2 in the presence of light to produce three isomers of C 5 H 11 Cl: This reaction replaces am one of the hydrogen?s of C 5 H 12 with a Cl. What arc the...
-
The cumulative incidence of myocardial infarction is 180.0 new cases per 10,000 persons among individuals with severe hypertension. In contrast, the cumulative incidence of myocardial infarction is...
-
Rundle Industries produces two electronic decoders, P and Q. Decoder P is more sophisticated and requires more programming and testing than does Decoder Q. Because of these product differences, the...
-
What financial statement, which can be reported with or separate from the income statement, includes income-related items that affect the balance sheet but are not included in net income?

Study smarter with the SolutionInn App


