Identify the reagents you would use to accomplish each of the following transformations: Br . -
Question:
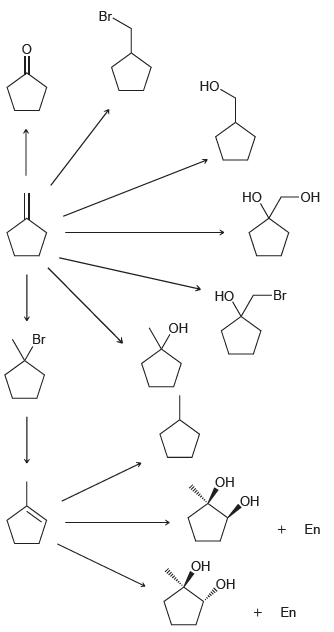
Transcribed Image Text:
Br Но. Но -ОН Br Но ОН Br он нин ОН En он Он En
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
1 03 2 DMS HBr NaOMe Br H...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the reagents you would use to accomplish the following transformation - Br Br
-
Identify the reagents you would use to accomplish the following transformation. CI
-
Identify the reagents that you would use to accomplish each of the following transformations: a. b. c. d. e. f. g. h. Br-
-
a. How could aspirin be synthesized, starting with benzene? b. Ibuprofen is the active ingredient in pain relievers such as Advil®, Motrin®, and Nuprin®. How could ibuprofen be...
-
Prove that if unions are done by size and path compression is performed, the worstcase running time is O(M(M,N)).
-
The tree-level invariant amplitude for the equivalent of Compton scattering in scalar QED was calculated in Eq. (7.6.54). Use this to calculate the unpolarized differential cross-section \(d \sigma /...
-
Although everyone makes mistakes, describe how history repeats itself when there are patterns of progress and regression in the provision of patient care. In forming your answer, consider how...
-
A major city in the northeast wants to establish a central transportation station from which visitors can ride buses to four historic landmarks. The city is arranged in a grid, or block, structure...
-
Sage Inc. experienced the following transactions for Year 1, its first year of operations: 1. Issued common stock for $80,000 cash. 2. Purchased $180,000 of merchandise on account. 3. Sold...
-
Payments to labor = WLC = 100 Payments to capital RKc = 150 Shoe: Sales revenue = PsQs = 120 Payments to labor = WVLS = 90 Payments to capital = RKS = 30 The production function for computers is:...
-
In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that proton transfer, and then predict the position of...
-
When a 65-year-old goes to the hospital, the part of Medicare that pays for the hospital bill is a. Part A. b. Part B. c. Part C. d. Part D.
-
Investors who purchased unregistered securities (nonexempt) are entitled to rescind the investment and are entitled to be paid back in full. a. True b. False
-
Bond A: A 9% bond with a 1,000 par value and coupons payable semi-annually is redeemable at maturity for 1,100. At a purchase price of 1120, the bond yields a nominal annual interest rate of i...
-
Assume a money market with safe and risky borrowers. The MFI's that is ready to lend money does not know who is safe and who is risky. However experience has led the MFI to know that 50% of the...
-
As a graduate from SDD-UBIDS who got employment at Kosmos energy in Accra at the Finance Directorate, the Director of Finance invited you to his office and asked you about the institution you did...
-
What element is one of the components of change management?
-
Which Billing Type requires project level billing set up of an item or schedule according to the contract?
-
Fill in the blank with an appropriate word, phrase, or symbol(s). When there are four candidates, the number of comparisons needed with the pairwise comparison method is ________________.
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
Make three-dimensional drawings of the following molecules, and predict whether each has a dipole moment. If you expect a dipole moment, show its direction. (a) H2C = CH2 (b) CHC13 (c) CH2C12 (d) H2C...
-
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO. :0: Nitromethane :O:
-
Calculate formal charges for the non-hydrogen atoms in the following molecules: (a) Diazomethane, (b) Acetonitrile oxide, (c) Methylisocyanide, H2C=N=N: part a H3C-NEC: part b H3C-CEN-o: part c
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


