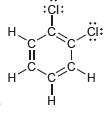
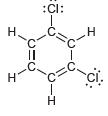
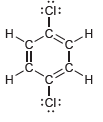
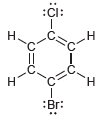
Identify which compounds below possess a molecular dipole moment and indicate the direction of that dipole moment:
Question:
a.
b.
c.
d.
Transcribed Image Text:
:Çi: ċi: .CI: Н II .C. Н .C. H. :ö: 一 一エ :Cl: Н. н .C. .C. di: Н °. CHO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a b c There is no mo...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds, predict which one has the higher molecular dipole moment, and explain your reasoning. (a) Ethyl chloride or ethyl iodide (b) 1-bromopropane or cyclopropane (c) Cis-2,...
-
Draw a Lewis structure for a compound with molecular formula C 4 H 11 N in which three of the carbon atoms are bonded to the nitrogen atom. What is the geometry of the nitrogen atom in this compound?...
-
What is the distinction between a bond dipole and a molecular dipole moment?
-
Last year, Intrepid Corporations tax return revealed the following items: dividends from 20 percent owned domestic corporations - $60,000; Gross income from services rendered - $300,000;...
-
During an expansion and at a peak, what forces are building up that will eventually help bring about a contraction in the economy?
-
Consider the model of a voluntary environmental agreement, in which there is a firm, a regulator, and a legislature. Benefits of abatement (in terms of reduced damage from pollution) are given by...
-
How can we use packaging to structure modeling artifacts?
-
Seasons Pizza recently hired additional drivers and as a result now claims that its average delivery time for orders is under 45 minutes. A sample of 30 customer deliveries was examined, and the...
-
3. Develop a plan for the Board which identifies some the problems and recommends a different approach to Innovation and CE in the organization, in particular emphasizing the importance of measuring...
-
Making a Decision as a Bank Loan Officer: Analyzing and Restating Financial Statements That Have Major Deficiencies (A Challenging Case) Julio Estela started and operated a small boat repair service...
-
When an acyl peroxide undergoes homolytic bond cleavage, the radicals produced can liberate carbon dioxide to form alkyl radicals: Using this information, provide a mechanism of formation for each of...
-
Fill in the worksheet in the following figure to estimate the relative effectiveness of these two different population control methods. The Chicken or the Egg Worksheet Find the differences in...
-
According to quantum mechanics, the energy of a harmonic oscillator is quantized. That is, it can take on any one of a certain set of values, given by where h is Plancks constant, equal to 6.62608...
-
What is the purpose of assistive equipment and their associated systems?
-
You can browse your account every few days to see which online payments have cleared and which have not. Most banks post your what in real time, although some still have a one or two day delay?
-
What would be the next step when an engagement team is unable to determine whether a GITC deficiency has been exploited?
-
Read the accounting case study, "Bridging the 'New Gap' in the Upcoming 10-K" Read the case study paying attention to the accounting guidelines, the likely changes the new revenue recognition...
-
Cory owns an apartment building. In addition to providing the utilities for his tenants, he also cleans the halls and utility room and picks up the trash. On what tax form must Cory report the...
-
The Transnational Business Inc. (TBI) purchased an asset that cost $60,000 on January 1, Year 1. The asset had a four-year useful life and a $10,000 salvage value . Required a. Determine the amount...
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
Arrange these compounds in order of decreasing SN2 reaction rate: CHCL CI Cl
-
Draw a free energy versus reaction progress diagram for a reaction that occurs in two steps with a relatively stable intermediate and in which the transition state for the second step is the...
-
Consider the free energy versus reaction progress diagram for the SN2 reaction shown in Figure 8.1. Does the transition state for this reaction have the C Cl bond less than half broke, approximately...
-
A consultant advises that a fund pays out $100,000 at the end of 5 years. Provided $12,000 is invested in the fund at the start of each year, beginning immediately and continuing up to and including...
-
Durco Automotive needs a $2 million balance in its contingency fund 3 years from now. The chief financial officer (CFO) wants to know how much to deposit now into Durco's high-yield investment...
-
3. Create a chart for gross margin less R&D and capital costs calculations. a. Calculate gross margin less R&D and capital costs for options A and B. b. Graph total gross margin less R&D and capital...

Study smarter with the SolutionInn App


