If an alkene is protonated and the solvent is an alcohol rather than water, a reaction takes
Question:
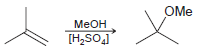
Transcribed Image Text:
OMe Меон [H,SOJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
T HOSO...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An alkene is treated with OsO4 followed by H2O2. When the resulting diol is treated with HIO4, the only product obtained is an unsubstituted cyclic ketone with molecular formula C6H10O. What is the...
-
Use Figure 4.5 to answer the following questions about the acid-catalyzed hydration of an alkene: a. How many transition states are there? b. How many intermediates are there? c. Which is more...
-
The difference in positive-charge distribution in an amide that accepts a proton on its oxygen or its nitrogen atom can be visualized with electrostatic potential maps. Consider the electrostatic...
-
Kenny operates a store, where he sells feed and other supplies to farmers. Heather purchases a $20,000 tractor from Kenny and pays Kenny with $18,000 in cash and $2,000 in corn. How much gross income...
-
What is meant by the completeness and soundness of Armstrong's inference rules?
-
For the generic state in exercise 1, calculate the entanglement entropy. Data From Exercise 1:- Consider the state \(\left|\chi_{1,2}^{a} ightangle=\left|\psi_{1,2} ightangle+a\left|\phi_{1,2}...
-
Distinguish the following data as discrete or continuous. Explain why. a. Temperature. b. Speed. c. Number of orders. d. Age. e. Weight. f. Price. g. Count.
-
Deborah purchases a new $30,000 car in 2014 to use exclusively in her business. If Deborah does not elect to expense in 2014 and holds the car until it is fully depreciated, how many years will this...
-
SHA E SI SINE THIS IS A TEST ASYL "NASA completed a record-breaking experiment last week, and it involved streaming an adorable cat video from 19 million miles away (about 80 times the distance...
-
Describe the actions taken by a thread library to context switch between user-level threads.
-
In this problem, you calculate the error in assuming that ÎH o R is independent of T for the reaction 2CuO(s) 2Cu(s) + O 2 (g). The following data are given at 25°C:
-
Using the reaction in the previous problem as a reference, propose a plausible mechanism for the following intramolecular reaction: [H,SO]
-
For the following exercises, write the equation of the line shown in the graph. x K amb 1112 mb nkn
-
Fleet Street Incorporated a manufacturer of high-fashion clothing for women, is located in South London in the UK. Its product line consists of trousers (28%), skirts (34%), dresses (15%), and other...
-
ABC Company is analyzing the purchase of new equipment. The following information is available: Initial investment Working capital needed now Equipment repair in 3 years Annual net cash inflows...
-
AndrIt iss dissolved 36.167gSn(C2H302), in enough distilled water to make 200.0mL of solution. Perform the following calculations regarding this solution: A. What is the molarity of this solution?...
-
By definition, an important aspect of culture is that it is living and living things evolve and change over time; they leave legacies. For this week, please discuss the connections you see between...
-
Peter Drucker indicated that it might be a waste of resources for organisations to invest in a training programme that is either unnecessary or inaccurate. Critically examine situations that may...
-
Use the circle graph to answer the questions. Students at Bayshore High School were polled to determine the type of music they preferred. There were 1960 students who completed the poll. Their...
-
Give codons for the following amino acids: (a) Th (b) Asp (c) Thr
-
Compound A, a hydrocarbon with M + = 96 in its mass spectrum, has the 13 C spectral data that follow. On reaction with BH 3 followed by treatment with basic H 2 O 2 , A is converted into B, whose 13...
-
Propose a structure for compound C, which has M + = 86 in its mass spectrum, an IR absorption at 3400 cm 1 , and the following 13 C NMR spectral data: Compound C Broadband-decoupled 13 C NMR: 30.2,...
-
Compound D is isomeric with compound C (Problem 13.61) and has the following 13C NMR spectral data. Propose a structure. Compound D Broadband-decoupled 13C NMR: 9.7, 29.9, 74.4, 114.4, 141.4 DEPT-90:...
-
Consider the control system in Figure where v(t) is a sinusoidal disturbance, v(t)=sin(t). Compute the absolute value of the sensitivity function at w = 1 rad/s as a function of K. How must K be...
-
ABC Corporation, a growing tech company, decides to raise capital by issuing convertible debt securities. Convertible debt allows bondholders the option to convert their debt into a predetermined...
-
Design Analog and Digital IIR Bandstop filter considering pass band edge1 20kHz, stop band edges 22 kHz & 38kHz, pass band edge2 40kHz, sampling frequency 100kHz. Additionally, draw corresponding...

Study smarter with the SolutionInn App


