Question: In Chapter 21, we will explore how nitriles can be converted into carboxylic acids. How would you use IR spectroscopy to monitor the progress of
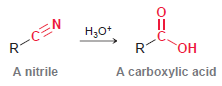
R-CEN A nitrile ,* C. A carboxylic acid
Step by Step Solution
3.41 Rating (173 Votes )
There are 3 Steps involved in it
The starting material has a cyano group C N and is ... View full answer

Get step-by-step solutions from verified subject matter experts


