In each of the following cases, determine if the base is sufficiently strong to deprotonate the terminal
Question:
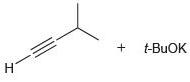
In each of the following cases, determine if the base is sufficiently strong to deprotonate the terminal alkyne. That is, determine whether the equilibrium favors formation of the alkynide ion.
(a)
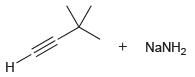
(b)
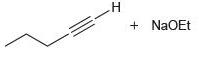
(c)
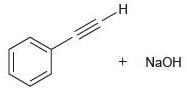
(d)
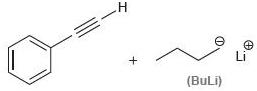
(e)
![]()
(f)
Transcribed Image Text:
NANH2 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a Yes NaNH 2 is strong enough of a base to deprotonate a terminal alkyne b No NaOEt is ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following cases, calculate the nominal exchange rate assuming purchasing power parity holds. a. A bottle of wine sells for $16 in the United States and 10 in France. b. A book sells...
-
In each of the following cases, imagine that the two reactants shown are allowed to reirct,in the presence of alcohol dehydrogenase. Tell whether the ethanol formed is chiral. If the ethanol is...
-
In each of the following cases draw the structure of an alkyl halide that will undergo an E2 elimination to yield only the indicated alkene. a. b. c. d. ? = E2 E2
-
Calculate the following limits at infinity. If necessary, use -inf and inf to denote -co and co. Use DNE if a limit does not exist. 1. lim e = 818 2. lim e = 8178 3. lim In(x) 24x In(x) =
-
A major department store chain is interested in estimating the mean amount its credit card customers spent on their first visit to the chain's new store in the mall. Fifteen credit card accounts were...
-
The following information is available for the years 2014, 2015 and 2015: The inventory valuations used above at the end of 2014 and at the end of 2015 were inaccurate. The inventory at 31 December...
-
You want to retire in 30 years. You are starting to invest in a growth income fund that promises an ambitious rate of $15 \%$. You can put in $\$ 200$ per month. How much will you have in 30 years?
-
Do Americans trust advertisements? The following table summarizes the results of a YouGov. com survey that asked Americans who see advertisements at least once a month how honest advertisements are....
-
This is a research to investigate the FAA maintainability requirements for aviation systems. Begin by considering the following statement from the United States General Accounting Office Testimony,...
-
Below are certain events that took place at Hazzard, Inc., last year: a) Collected cash from customers b) Paid cash to repurchase its own stock c) Borrowed money from a creditor d) Paid suppliers for...
-
Which international water problems have been most amenable to management? Least amenable?
-
The pK a of CH 3 NH 2 is 40, while the pK a of HCN is 9. (a) Explain this difference in acidity. (b) Can the cyanide anion (the conjugate base of HCN) be used as a base to deprotonate a terminal...
-
Write the structural formula for each of the following compounds. a. 3-ethyl-1-pentene b. 1,1,2,2-tetraphenylethane c. 1-phenyl-2-butanone d. Cyclopentanone
-
Why does the exact same model of refrigerator cost more in Germany than it does in the U.S.?
-
The fact remains that it is cost-effective and efficient for firms to source their supply chains from across the globe. This phenomenon is known as?
-
a. The U.S. dollar derives its value from what? That is, what "backs" U.S. currency? b. What is the term used to describe the type of money used in the United States today? c. What two other types of...
-
Sheridan Manufacturing purchased a new building with the surrounding land. The price paid was $2,400,000, including the commission charged on the sale. An appraisal of the land and building at the...
-
What does The British north America act of 1867 contains?
-
Mr. and Mrs. Chan want to buy furniture that has a cash price of $3450. On the installment plan, they must pay 25% of the cash price as a down payment and make six monthly payments of $437. (a) What...
-
A handrail, which weighs 120 N and is 1.8 m long. was mounted to a wall adjacent to a small set of steps (Figure P4.26). The support at A has broken, and the rail has fallen about the loose bolt at 8...
-
What product(s) would you expect from reaction of (S)-3-chloro-3-methyloctane with acetic acid? Show the stereo chemistry of both reactant and product.
-
Among the numerous examples of SN1 reactions that occur with incomplete racemization is one reported by Winstein in 1952? The optically pure tosylate of 2, 2-dimethy-1-phenyl-1-propanol ([?] D = ?...
-
Assign configuration to the following substrate, and show the stereo chemistry and identity of the product you would obtain by SN1 reaction with water (reddish brown =Br):
-
I. Analyze the geometric construction of the systems shown as in figures. A Figure 1-1 E Figure 1-2 Figure 1-3 bisg ela ad bloode vuilidizel Figure 1-4
-
structural engineering course, i need it asap please. thanks A. Calculate the reactions of the two-span frame shown in figure Figure 4-1 2.5kN/m (a) D E F A B 2m 2m 4m 0 4m B. Calculate member-end...
-
As a hired structural engineering consultant, you were requested to evaluate a four-story building that is damaged by a Magnitude 6 earthquake. The client has no background in any civil/structural...

Study smarter with the SolutionInn App


