Question: In the next chapter, we will learn a method for preparing alkynes (compounds containing C ¡ C triple bonds). In the following reaction, a dihalide
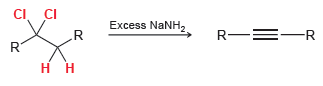
CI. CI .R Excess NaNH, R-=-R R. R.
Step by Step Solution
3.41 Rating (173 Votes )
There are 3 Steps involved in it
CI CI... View full answer

Get step-by-step solutions from verified subject matter experts


