Question: Match each compound with the appropriate spectrum. a. b. c. d. .CI OH Br ZHN
Match each compound with the appropriate spectrum.
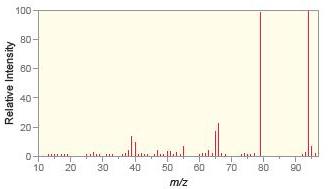
a.
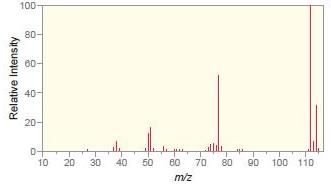
b.
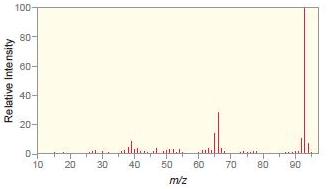
c.
d.
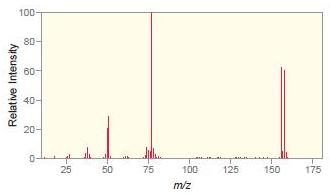
.CI OH Br ZHN
Step by Step Solution
3.43 Rating (156 Votes )
There are 3 Steps involved in it
a ... View full answer

Get step-by-step solutions from verified subject matter experts


