Methylmagnesium bromide reacts rapidly with ethylene oxide, it reacts slowly with oxetane, and it does not react
Question:
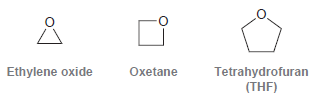
Explain this difference in reactivity.
Transcribed Image Text:
Oxetane Ethylene oxide Tetrahydrofuran (THF)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Ethylene oxide has a high degree of ring strain and readily functions as an electrophile in an S ...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write an equation for the reaction, if any, of p-bromobenzaldehyde with each of the following reagents, and name the organic product. a. Methylmagnesium bromide, then H3O+ b. Methylamine (CH3NH2) c....
-
A compound reacts with methylmagnesium bromide followed by acidification to form the product with the following 1H NMR spectrum. Identify the compound. 6 (ppm) 2 frequency
-
Ethylene oxide, C2H4O, decomposes when heated to give methane and carbon monoxide. C2H4O(g) CH4(g) + CO(g) The following kinetic data were observed for the reaction at 688 K: Find the rate law and...
-
DBU Systems manufactures testing equipment for the communications industry. In developing a new device for maritime communication, the design group has estimated the following unit costs. Metal...
-
Parent Co. purchases 100% of Son Company on January 1, 20X1 when Parent's retained earnings balance is $520,000 and Son's is $150,000. During 20X1, Son reports $15,000 of net income and declares...
-
Sacred Heart Hospital has the following receivables amounts, listed by age: a. Complete the aging schedule by filling in the percentage of total value column. b. Interpret your results. c. Using...
-
Consider a deterministic cash flow stream \(\left(x_{0}, x_{1}, x_{2}, \ldots, x_{n} ight)\) with all positive flows. Let \(\mathrm{PV}(r)\) denote the present value of this stream at an interest...
-
A camera of mass 240 g is mounted on a small tripod of mass 200g, assuming that the mass of the camera is uniformly distributed and that the line of action of the weight of the tripod passes through...
-
use the formula approach or calculator approach, answer the following questions: 19. Suppose you want to borrow $20,000 for a new car. You can borrow at 8% per year, compounded monthly. If you take a...
-
A dc generator has an internal resistance R ohms and develops an open circuit voltage of V volts (Fig. 2.10). Find the value of the load resistance for which the power delivered by the generator...
-
The Williamson ether synthesis cannot be used to prepare tert-butyl phenyl ether. a. Explain why this method cannot be used in this case. b. Suggest an alternative method for preparing tert-butyl...
-
Identify the reagents necessary to accomplish each of the following transformations. >
-
Rockland Corporation earned net income of R$300,000 in 2015 and had 100,000 ordinary shares outstanding throughout the year. Also outstanding all year was R$800,000 of 10% bonds, which are...
-
How do advanced computational fluid dynamics (CFD) techniques facilitate the analysis and optimization of complex fluid flow phenomena in industrial-scale reactors, considering turbulent flow...
-
How do advanced spectroscopic techniques such as NMR spectroscopy, FTIR spectroscopy, and X-ray diffraction enable the elucidation of molecular-level interactions and structural changes in complex...
-
Julie is the owner / operator of a massage parlor in Carmel. Last year she earned $ 4 0 0 , 0 0 0 in total revenue. Her explicit costs were $ 2 0 0 , 0 0 0 . During the year she received offers to...
-
Attached are the two copies of the statement. Your assignment is to read the statement (it is not too long) very carefully, and in one-page maximum, write an assessment of the stakeholder vs...
-
prepare the script between client and financial planner = I'm not prepared to move forward with the portfolio you've presented right now because I'm afraid that Donald Trump might take over the U.S....
-
Suppose that the probability that a mechanic fixes a car correctly is 0.9. Determine the odds against the mechanic fixing a car correctly.
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
In step 6 of fatty-acid biosynthesis (Figure), acetoacetyl ACP is reduced stereo specifically by NADPH to yield an alcohol. Does hydride ion add to the Si face or the Re face of acetoacetylACP?...
-
In step 7 of fatty-acid biosynthesis (Figure), dehydration of a ?-hydroxy thioester occurs to give trans-crotonyl ACP. Is the dehydration a syn elimination or an anti elimination? CH3SCOA Acetyl CoA...
-
In step of fatty-acid biosynthesis (Figure), reduction of Trans-crotonyl ACP gives butyryl ACP. A hydride from NADPH adds to C3 of the crotonyl group from the Re face, and Protonation on C2 occurs on...
-
The first video I have for your discussion is "Shock and Awe:The Story of Electricity" Part 1 is called "Spark"It runs from 0:00 to about 58 min, on the following link....
-
Find the present value of a 1 0 - year annuity of $ 8 0 0 0 per annum, given the interest rate of 6 % for the first year, 7 % for the second year, 8 % for the third year, 9 % for the fourth year, and...
-
The management accounting team of XYZ plc also came up with some questions and requested you to explain/answer them for the upcoming board meeting: 1. A chain of XYZ plc, BHealthy Ltd is a wholesale...

Study smarter with the SolutionInn App


