Nitrogen and sulfur are neither in the same row nor in the same column of the periodic
Question:
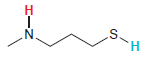
Transcribed Image Text:
.S. H. エーZ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
A proton connected to a sulfur atom will be more acidi...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Ammonium sulfate is used as a nitrogen and sulfur fertilizer. It is produced by reacting ammonia with sulfuric acid. Write the balanced equation for the reaction of gaseous ammonia with sulfuric acid...
-
Which of the hydrogen atoms shown below is more acidic? Explain your answer. A
-
The diagram that follows shows the highest-energy occupied MOs of a neutral molecule CX, where element X is in the same row of the periodic table as C. (a) Based on the number of electrons, can you...
-
In Exercises, find the limit. lim (4x- x 16x - x
-
One area that we haven't looked at yet is payroll. Can you get the documentation from Karina for payroll and then complete a "bridge work paper" like you did for the Revenue and Purchasing cycles? As...
-
During the month of April, you conduct art seminars for children. On April 1, you borrow $500 from your parents, buy $400 of supplies, and pay $50 for advertising on the radio and $25 to rent a room...
-
CD Real Estate specializes in residential real estate services in the state of California. To complement the experience and local market knowledge of its licensed realtors, CD Real Estate wants to...
-
The Wear ever Shoe Company is going to open a new branch at a mall, and company managers are attempting to determine how many salespeople to hire. Based on an analysis of mall traffic, the company...
-
A concave spherical mirror has a focal length of 6.00 cm. If an object is placed at a distance of 24.0 cm from the mirror, calculate the image distance and characteristics of the image including...
-
A tall standpipe with an open top (see figure) has diameter d = 2.2 m and wall thickness t = 20 mm. (a) What height h of water will produce a circumferential stress of 12 MPa in the wall of the...
-
In each compound below, two protons are clearly identified. Determine which of the two protons is more acidic. (a) (b) (c) (d) N' C-
-
In each compound below, two protons are clearly identified. Determine which of the two protons is more acidic. (a) (b) (c) (d) (e) (f) -N
-
Determine whether each function is one-to-one. If it is, find the inverse. {(-8, 6), (-4, 3), (0, 6), (5, 10)}
-
Why was Novos P/E ratio so much smaller than its key competitors P/E s ? What were the implications for Novos ability to compete in world markets?
-
Explain the concept of mildly segmented capital markets.
-
What are Yankee, samurai, or bulldog bonds?
-
What is the difference between the euro bond market and the Eurobond market?
-
Define what is meant by the offshore or Eurobond market.
-
A company assigns overhead cost to completed jobs on the basis of 150% of direct labour cost The job cost sheet for Job 313 shows that $12,000 in direct materials was used on die job and that $16,000...
-
A stock has had returns of 8 percent, 26 percent, 14 percent, 17 percent, 31 percent, and 1 percent over the last six years. What are the arithmetic and geometric average returns for the stock?
-
The halogen atom of an alkyl halide can be replaced by the hydrogen atom bonded to tin in tributyltin hydride (Bu3SnH). The process, called dehalogenation, is a radical reaction, and it can be...
-
Write a mechanism that accounts for the following reaction. The hydrogen atom bonded to tin in tributyltin hydride is readily transferred in radical mechanisms. Br PhCO22, BuSnH (Major)
-
Molecular orbital calculations can be used to model the location of electron density from unpaired electrons in a radical. Open the molecular models on the book's website for the methyl, ethyl, and...
-
Assume that in 2023, Bobby had a vacant lot that was given to him as a gift from his father that had an FMV of $40,000 and an adjusted basis of $15,000.
-
Show the adjusting entries that you would make given the additional data below. In addition to the formal journal entry, please also indicate the type of adjusting entry that is being made, ...
-
I need one to three paragraphs written about the awk command in unix. Everything should be in a power point presentation. Make sure it contains the following: Command name Description How to use the...

Study smarter with the SolutionInn App


