Predict the major and minor product for each of the following E2 reactions: a. b. c. d.
Question:
a.
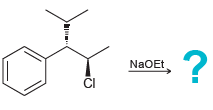
b.
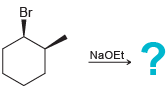
c.
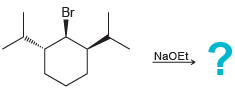
d.
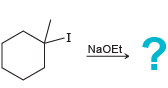
e.
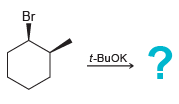
f.
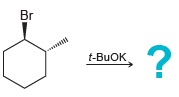
Transcribed Image Text:
? NaOEt ČI Br NaOEt,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a b c...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the dehydrohalogenation product(s) that result when the following alkyl halides are heated in alcoholic KOH. When more than one product is formed, predict the major and minor products. (a)...
-
Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one product is expected, label the major and minor products. (a) 2-methylbutan-2-ol (b)...
-
Identify the major and minor product(s) that are expected for each of the following reactions: a. b. c. d. e. f. g. h. i. j. k. l. m. n. OTs Naci DMSO NaOH
-
using System; using System.Collections.Generic; using System.ComponentModel; using System.Data; using System.Drawing; using System.Linq; using System.Text; using System.Threading.Tasks; using...
-
A dry quartz sand has a density of 1.68 Mg/m3. Determine its density when the degree of saturation is 75%. The density of solids for quartz is 2.65 Mg/m3.?
-
The transfer function \[H(s)=\frac{\kappa}{\left(s^{2}+1.4256 s+1.23313 ight)(s+0.6265)}\] corresponds to a lowpass normalized Chebyshev filter with passband ripple \(A_{\mathrm{p}}=0.5\)...
-
The Hull White model is \[d X_{t}=a(t)\left(b(t)-X_{t} ight) d t+\sigma(t) d W_{t}\] In this problem take \(a(t)=\theta_{1} t, b(t)=\theta_{2} \sqrt{t}\) and \(\sigma(t)=\theta_{3} t\) where...
-
An off-duty, out-of-uniform police officer and his son purchased some food from a 7- Eleven store and were still in the parking lot when a carload of teenagers became rowdy. The officer went to speak...
-
You will deposit $2,000 at the end of each of next 3 years. If the interest rate is 10% (annual compounding), how much will you have accumulated in 26 years?
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Compare integrated pest management to integrated waste management. How does each reduce potential damage to the environment?
-
How does the system of integrated waste management depicted in Figure 23.10 compare to a natural ecosystem?
-
Show that if |x + 3| < 1/2, then |4x + 13 | < 3.
-
Write a research essay/paper that addresses your point of view on a current ethical topic in the news. Your paper should include convincing evidence. Look in the newspaper or news website (CNN, NBC,...
-
Effective followers assist in productive leadership behavior in the same way that effective leaders produce good followers in their workforce. I have less score on this assessment; therefore, to be a...
-
1. What are the 3 types of communication according to mode? 2. How do you differentiate the types of communication according to mode? 3. How can visual communication enhance the message conveyed by a...
-
Write a 1 short paragraph story in the form of a list of New Year's resolutions. dont use Chat AI ?
-
Explain Netflix's early years and its transition from DVD sales to DVD rentals. Lastly, what were the results and experiences or lessons learned by Netflix from this change?
-
What changes during a chemical reaction?
-
a. Why does the Wi-Fi Alliance release compatibility testing profiles in waves instead of combining the entire standards features initially? 27a1.) An 802.11ac Wi-Fi compatibility testing profile...
-
How would you carry out the following conversions? More than one step may be needed is some instances. CI RCH RCHCH3 RCH2CH3 RCH2CH2OH R-C=CH R-C C-CH3 RCH=CH2 R-C- RCCH3
-
Hydrocarbon A has the formula C 9 H 12 and absorbs 3 equivalents of H 2 to yield R, C 9 H 18 , when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H 2 SO 4 in the presence of...
-
How would you carry out the followingreactions? (a) CCH-CCH3 CH3CH2C=CH () H2C%3CH CH3CH2CH2CHO .3- (c) 3C (d) CH . CH 7, 22H (e) CH3CH2C=CH (f) CH3CH2CH2CH2CH=CH2 CHCH2CH2CH2C%CH (2 steps)
-
Hall & Company issues $ 1 0 0 , 0 0 0 , 0 0 0 in 6 % bonds with detachable warrants of 1 0 warrants for each $ 1 , 0 0 0 bond. The bonds are issued at 9 9 . Record the issuance under the following...
-
Assume that hein acquired Dolan Development last year. Hein recorded the following intangible assets on the date of acquisition: Goodwill: $ 1 , 5 0 0 , 0 0 0 Dolan Development trademark: $ 6 0 0 , 0...
-
What is the total amount of interest that will be paid of the loan is carried to term with no extra payments made building cost 3 3 0 0 0 0 . loan started im Dec 2 0 2 3 worh a 7 . 5 % interest rate...

Study smarter with the SolutionInn App


