How would you carry out the followingreactions? (a) CCH-CCH3 CH3CH2C=CH () H2C%3CH CH3CH2CH2CHO .3- (c) 3C (d)
Question:
How would you carry out the followingreactions?
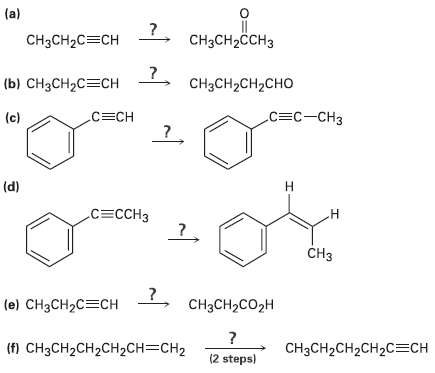
Transcribed Image Text:
(a) CнзCH-CCH3 CH3CH2C=CH (ы) снзсH2C%3CH CH3CH2CH2CHO .С3с-сНз (c) С3CН (d) н СЕСCHЗ .н CHз 7, снасн2со2H (e) CH3CH2C=CH (f) CH3CH2CH2CH2CH=CH2 CHзCH2CH2CH2C%ЕCH (2 steps)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
b c CH3CHCCH CH3CHCCH CCH HO HSO4 HgSO4 1 BH3 THF ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
At December 31, 2014, Torrealba Company reported the following as plant assets. During 2015, the following selected cash transactions occurred. April 1 Purchased land for $1,200,000. May 1 Sold...
-
A set of ten jobs must be scheduled at an integrated machining center that performs a number of metal-cutting operations on components for complex assemblies. These jobs and their processing times...
-
Find Zab in the four way power divider circuit in Fig. 2.135. Assume each element is 1(. Figure 2.135 For Prob. 2.75. bo
-
You are provided with the profit centre responsibility accounting reports for Volantas Department Stores. Required (a) Explain the relationship between the three profit responsibility reports for...
-
Maria Ochoa receives two new credit cards on May 1. She had solicited one of them from Midtown Department Store, and the other arrived unsolicited from High-Flying Airlines. During the month of May,...
-
- (i). [7 marks] Given g(1) = 1, show the solution of g(n) = g(n - 1) + 4n is O(n). - (ii). [7 marks] Analyze the time complexity of MAXSUBARRAYSUM algorithm. Algorithm MAXSUBARRAYSUM (A, l, h) 1:...
-
Automatic control of helicopters is necessary because, unlike fixed-wing aircraft which possess a fair degree of inherent stability, the helicopter is quite unstable. A helicopter control system that...
-
Hydrocarbon A has the formula C 9 H 12 and absorbs 3 equivalents of H 2 to yield R, C 9 H 18 , when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H 2 SO 4 in the presence of...
-
Occasionally, chemists need to invert the stereochemistry of an alkene?that is, to convert a cis alkene to a trans alkene, or vice versa. There is no one-step method for doing an alkene inversion,...
-
For the following exercises, find all complex solutions (real and non-real). 3x 3 4x 2 + 11x + 10 = 0
-
The table below summarizes asset allocations recommended by Blackrock and Charles Schwab to three different types of investors: Conservative investors with high risk aversion, moderate investors with...
-
Find (f+g)(-2) when f(x)=x+2 and g(x) = x - 1.
-
What is the organizational impact of a laissez faire leadership style ? Discuss in detail
-
How to improve vaccination rates has been a publicized topic over the last few years. There is controversy on the safety and unknown adverse negative externalities. A lack of trust in our government,...
-
need help resolving this on excel please 1 The Company WSpectacles is a firm that organizes concerts, festivals and spectacles in Paris. In particular, WSpectacles focuses on events for young people,...
-
Let R be the event of randomly selecting a senator and getting a Republican, and let D represent the event of randomly selecting a second different senator and getting a Democrat. Use words to...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
Consider the precipitation reaction: What volume of 0.175 M Na3PO4 solution is necessary to completely react with 95.4 mL of 0.102 M CuCl 2 ? 2 Na3PO4(aq) + 3 CuCl(aq) Cu3(PO4)2 (s) + 6 NaCl(aq)
-
Draw a Lewis structure for each species. (a) N2H4 (b) N2H2 (c) (CH3)2NH2CI (d) CH3CN (e) CH3CHO (f) CH3S(O)CH3 (g) H2SO4 (h) CH3NCO (i) CH3OSO2OCH3 (j) CH3C(NH)CH3 (k) (CH3)3CNO
-
Draw a Lewis structure for each compound. Include all nonbonding pairs of electrons. (a) CH3COCH2CHCHCOOH (b) NCCH2COCH2CHO (c) CH2CHCH(OH)CH2CO2H (d) CH2CHC(CH3)CHCOOCH3
-
Draw a line-angle formula for each compound in Problem 1-26. In problem (a) CH3COCH2CHCHCOOH (b) NCCH2COCH2CHO (c) CH2CHCH(OH)CH2CO2H (d) CH2CHC(CH3)CHCOOCH3
-
In what ways do organizations integrate sustainability principles and circular economy concepts into their innovation strategies, including cradle-to-cradle product design, resource recovery...
-
On December 31, 2022, Skysong Inc. owns a machine with a carrying amount of $824,000. The original cost and accumulated depreciation for the machine on this date are as follows: Machine $1,400,000...
-
discuss the following terms Concepts of Motivation at work place 2) Six Real life situations of Motivation at work place 3) Benefits observed of Motivation at work place 4) Limitations observed of...

Study smarter with the SolutionInn App


