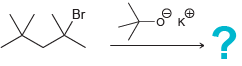
Predict the major product for each of the following reactions: a. b. c. d. e. f. g.
Question:
a.
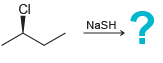
b.
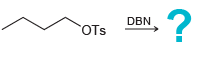
c.
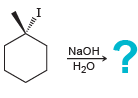
d.

e.
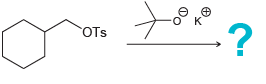
f.
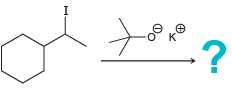
g.
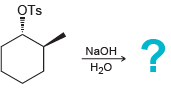
h.
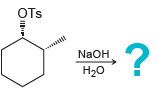
i.
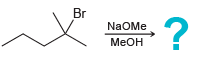
j.
Transcribed Image Text:
CI :? NaSH DBN OTs
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a b c...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product(s) and mechanism(s) for reaction between 2-bromobutane (sec-butyl bromide) and each of the reagents in Problem 42. (a) KCl in DMF (b) KI in DMF (c) KC1 in CH3NO2 (d) NH3 in...
-
Predict the major product(s) and mechanism(s) for reaction between 2-bromo-2-methylpropane (tert-butyl bromide) and each of the reagents in Problem 42. (a) KCl in DMF (b) KI in DMF (c) KC1 in CH3NO2...
-
Predict the major product(s) for each of the following reactions: 1) Hg(OAC)2, -0 2) NABH, ? - O, NaOH, cold Br2 H20 Pt
-
Use the data in Exercise 13.28.To familiarize yourself with recursive least squares, estimate the savings functions for 19701981, 19701985, 19701990, and 19701995. Comment on the stability of...
-
Read article "The Organization of Personality" by H. J. EYSENCK and write a reaction paper
-
Indicate whether each of the following statements is true or false: a. The cash basis of accounting recognizes revenues when they are received. b. In the cash basis of accounting, there is no such...
-
Show that the circulation of a free vortex for any closed path that encloses the origin is \(\Gamma\).
-
Elwood Inc. purchased computer equipment on March 1, 2012, for $36,000. The computer equipment has a useful life of 10 years and a salvage value of $3,000. For tax purposes, the MACRS class life is 5...
-
Belloq Inc. was a startup meal kit delivery service with its headquarters in Massachusetts. It sold meal kits to customers over the Internet, packaged them with instructions for preparation, and...
-
A pulley is keyed to a shaft midway between two anti-friction bearings. The bending moment at the pulley varies from 170 N-m to 510 N-m and the torsional moment in the shaft varies from 55 N-m to 165...
-
When 2-bromo-2-methylhexane is treated with sodium ethoxide in ethanol, the major product is 2-methyl-2-hexene. a) Draw the mechanism of this reaction. b) What is the rate equation of this reaction?...
-
What are the main arguments for and against the United States developing additional nuclear power plants to provide us with electricity over the next several decades? Which perspective do you find...
-
Following are selected December 31 account balances of Kosciusko, Inc. prior to the preparation of year-end adjusting journal entries: Property, Plant &...
-
Management accounting is a mid-way between financial accounting and cost accounting. Elucidate.
-
Name the accounting principle involved/violated/affected in the following: (i) The firm changes method of depreciation. (ii) The firm does not consider unused stationery as asset. (iii) The firm...
-
Explain the following: (a) Management accounting is an extension of financial accounting. (b) Management accounting assists in corporate planning process.
-
Name the 5 major areas in which different accounting policies can be adopted by business enterprises. What is the requirement of the relevant accounting standard in this regard?
-
How would you analyse the financial position of a company from the point of view of (a) an investor, (b) a creditor, and (c) a financial executive of the company?
-
Prove that lim In x x0+ = -0.
-
Using thermodynamic data from Appendix 4, calculate G at 258C for the process: 2SO 2 (g) + O 2 (g) 88n 2SO 3 (g) where all gases are at 1.00 atm pressure. Also calculate DG8 at 258C for this same...
-
Nitriles, RC N, undergo a hydrolysis reaction when heated with aqueous acid. What is the structure of the compound produced by hydrolysis of propane nitrile, CH3CH2C N, if it has IR absorptions at...
-
The amount of energy required to spin-flip a nucleus depends both on the strength of the external magnetic field and on the nucleus. At a field strength of 4.7 1 rf energy of 200 MHz is required to...
-
Calculate the amount of energy required to spin-flip a proton in a spectrometer operating at 300MHz. Does increasing the spectrometer frequency from 200 to 300 MHz increase or decrease the amount of...
-
a) Give one reason why local Pakistan bus businesses could benefit from Pakistan's crisis. b) Give one reason why local bus businesses could be hurt by the Rupee crisis.
-
A P/E multiple could be high relative to the S&P 500 Index because its high growth rate justifies it. You notice a stock that has an assumed growth rate of 5.43% with a dividend payout ratio of 64.9%...
-
What would be the volume ( cm 3 ) of a piece of Styrofoam if it had a mass of 3 5 0 g , no matter its shape? Show calculation.

Study smarter with the SolutionInn App


