Predict the stereochemical outcome for each of the following E2 reactions. In each case, draw only the
Question:
a.
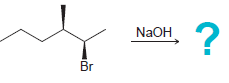
b.
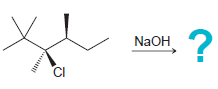
Transcribed Image Text:
? NaOH Br NaOH CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a b...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use your answers to the preceding problem to predict the stereochemical outcome of the addition of bromine to maleic acid and to fumaric acid. (a) Which dicarboxylic acid would add bromine to yield a...
-
Predict the stereochemical outcome of radical bromination of the following alkanes: (a) (b) (c) (d)
-
Predict the major product for each of the following E2 reactions: a. b. c. d. NaOH Br NaOH Br
-
What is the "ALDI Way" and what was its quest? Take out costs; eliminate complexity Survival; make enough money to pay overhead costs Increase market share; find more prospective customers Build...
-
A Christian Philosophical journey by Schenck kenneth In a Word document, respond to all the following questions: How do I know what is true? Can I trust my senses? Can I trust my mind? Does God...
-
_______________________ It is not necessary to have equal sample sizes for the paired t test.
-
Why should operations be described by verbs?
-
1. Compute he financial ratios discussed in the chapter for Wholesome Foods for 2012 and 2013. 2. Prepare a cash flow statement for the firm for 2012 and 2013.
-
The total cost (in dollars) of manufacturing x auto body frames is C(x)=20,000+ 700x. (A) Find the average cost per unit if 250 frames are produced. (B) Find the marginal average cost at a production...
-
The statement of financial position of Bharwani Traders on 31 October 204 is shown below. On 1 November 204 the following transactions took place: A cheque for $3 000 was paid to a credit supplier. A...
-
Some energy experts refer to the Great Plains states as the Saudi Arabia of wind power. Explain what the reference means.
-
Japan wishes to make use of solar power, but it does not have extensive tracts of land for building large solar power plants. Which solar technology do you think is best suited to Japan's needs? Why?
-
"Taxes on tobacco and alcohol are nearly perfect taxes. They raise lots of revenue and discourage smoking and drinking." a. To what extent are the two effects inconsistent? b. How is the burden of an...
-
Sandhill Beauty Corporation manufactures cosmetic products that are sold through a network of sales agents. The agents are paid a commission of 21% of sales. The income statement for the year ending...
-
In the current year, Sandra rented her vacation home for 75 days, used it for personal use for 22 days, and left it vacant for the remainder of the year Her Income and expenses before allocation are...
-
A mass 100 g hangs from the spring scale. When a professor moves the scale up with acceleration 5 m/s. What is the reading of the spring scale?
-
How will the frequency of the pendulum change, if we increase its length four times?
-
Two items are omitted from each of the following summaries of balance sheet and income statement data for two corporations for the year 2022, Cullumber Company and Ayayai Enterprises. Determine the...
-
A quinella bet consists of selecting the first- and second-place winners, in any order, in a particular event. For example, suppose you select a 25 quinella. If 2 wins and 5 finishes second, or if 5...
-
Rewrite Programming Exercise 7.5 using streams. Display the numbers in increasing order. Data from Programming Exercise 7.5 Write a program that reads in 10 numbers and displays the number of...
-
The following reaction involves a conjugate addition reaction followed by an intra molecular Claisen condensation. Write both steps, and show theirmechanisms. CO2CH3 (CH3i>CuLi -CEC-CO2CH3 H
-
The following reaction involves two successive intra molecular Michael reactions. Write both steps, and show theirmechanisms. Nat "OEt Ethanol
-
The following reaction involves an intra molecular aldol reaction followed by a ret ro aldol-like reaction. Write both steps, and show theirmechanisms. O2Et CO2Et Na* "OEt Ethanol
-
A project costs $80 MM, with 25% of the investment int in year 1 and the balance in year 2. The plant operates atates at 50% capacity in year 3 and then full capacity for the next next 20 years. The...
-
What is organizational Behaviour the study of? How is social sciences related to organizational behavior? What is the difference between social science and organizational behavior? What is the...
-
Describe the nature of organization change, including forces for change and planned versus reactive change

Study smarter with the SolutionInn App


