Predict whether each of the following carbocations will rearrange. If so, draw the expected rearrangement using curved
Question:
a.
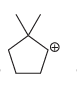
b.
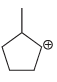
c.
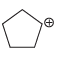
d.
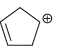
e.
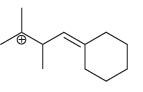
f.
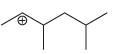
g.
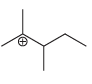
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a b c This carbocation is seco...View the full answer

Answered By

Michael Mulupi
I am honest,hardworking, and determined writer
4.70+
72+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict whether each of the following molecules is polar or non-polar: (a) IF (b) CS2 (c) SO3 (d) PCl3 (e) SF6 (f) IF5
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict whether each of the following compounds is molecular or ionic: (a) B2H6 (b) CH3OH (c) LiNO3 (d) Sc2O3 (e) CsBr (f) NOCl (g) NF3 (h) Ag2SO4.
-
The inequality describes the range of monthly average temperatures T in degrees Fahrenheit at a certain location. (a) Solve the inequality. (b) If the high and low monthly average temperatures...
-
Read the case "The Boys Versus Corporate" and write a short paper related to cultures
-
The distribution of service times at the drive-up window of the Kermit Roosevelt Bank is bell shaped, with a mean of 3.5 minutes and a standard deviation of 1.1 minutes. What percentage of service...
-
Develop an analytical model for determination of the CTE, \(\alpha\), for a randomly oriented continuous fiber composite in terms of fiber and matrix properties and volume fractions. Assume that the...
-
Jones, wishing to retire from a business enterprise that he had been conducting for a number of years, sold all of the assets of the business to Jackson Corp. Included in the assets were a number of...
-
Do you think that people under the age of 18 should be required to wear protective helmets when skateboarding, in-line, bicycling, snowboarding, or skiing? Why or why not.
-
Schrand Aerobics, Inc., rents studio space (including a sound system) and specializes in offering aerobics classes. On January 1, 2019, its beginning account balances are as follows: Cash,...
-
Draw curved arrows for each step of the following mechanism: : : : 0: . : : -O: : - : : : : H20 -
-
Consider the following reaction: The following rate equation has been experimentally established for this process: Rate = k [HO - ] [CH 3 CH 2 Br] The energy diagram for this process is shown below:...
-
Show that less than complete specialization in production leads to a lower level of welfare than complete specialization.
-
Pricing affects the positioning of a product offering. Explain the process of determining the pricing strategy.
-
Following a sharp increase in the price of energy, the overall price level is most likely to rise in the short run: A. and remain elevated indefinitely unless the central bank tightens. B. but be...
-
Writing a strangle (advanced). Assuming that Allied-Lyons would write a strangle as a speculative strategy, rather than a straddle as in problem 7, would you consider it to be more or less...
-
Comment on the importance of value creation product strategies for transactional, consultative, and strategic alliance buyers.
-
Who are the primary users of financial accounting information?
-
If a firm uses n inputs (n > 2), what inequality does the theory of revealed cost minimization imply about changes in factor prices (w i ) and the changes in factor demands (x i ) for a given level...
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Compound B, an isomer of A (Problem 20.47), is also soluble in dilute HCl. The IR spectrum of B shows no bands in the 3300-3500-cm-1 region. The broadband proton-decoupled 13C spectrum of B is given...
-
Compound C (C9H11NO) gives a positive Tollens' test (can be oxidized to a carboxylic acid) and is soluble in dilute HCl. The IR spectrum of C shows a strong band near 1695 cm-1 but shows no bands in...
-
Show how you might prepare each of the following amines through reductive amination: (a) (b) (c) NH2 CH3 CH3
-
1. Explain what the "Deinstitutionalization Mandate" is. This should be done in at least two paragraphs. 2. In another two paragraphs, discuss how status offenses are connected to the...
-
You are the owner of a very small business that sells gourmet coffee. You sell only one product, a 12-ounce bag of whole-bean French roast coffee. You sell each bag of coffee for $14 each, but due to...
-
Find the instantaneous rate of change of the function f(t)=3+5t when t=-3.

Study smarter with the SolutionInn App


