Propose a mechanism of formation for each of the following products: ETOH Heat OEt OTs OEt
Question:
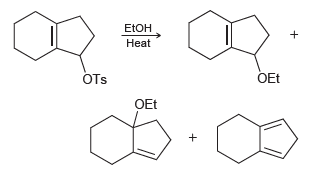
Transcribed Image Text:
ETOH Heat OEt OTs OEt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
COTS COTS Tad C...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When an acyl peroxide undergoes homolytic bond cleavage, the radicals produced can liberate carbon dioxide to form alkyl radicals: Using this information, provide a mechanism of formation for each of...
-
When ethoxybenzene is treated with a mixture of nitric acid and sulfuric acid, two products are obtained each of which has the molecular formula C 8 H 9 NO 3 . (a) Draw the structure of each product....
-
Propose a mechanism for each reaction. (a) (b) Ht H3C CH3 OH H SOA Ph Ph Ph Ph
-
The cable is subjected to a uniform loading of w = 60 kN/m. Determine the maximum and minimum tension in cable. -100 m- 12 m
-
Alcohol is a central nervous system (CNS) depressant used by about half of all Americans. Alcohol's effect on the body is measured by the blood alcohol concentration (BAC). The higher the BAC is the...
-
According to an almanac, 70% of adult smokers started smoking before turning 18 years old. (a) Compute the mean and standard deviation of the random variable X, the number of smokers who started...
-
How do you manage risk in the Spiral Model?
-
A negotiable promissory note executed and delivered by B to C passed in due course and was indorsed in blank by C, D, E, and F. G, the present holder, strikes out Ds indorsement. What is the...
-
The carrying case would be sold to the end consumer for $ 4 9 . 9 9 . Lisa will not sell directly to the consumer but will use a wholesaler who will sell to a retailer ( e . g . , university...
-
Juarez Inc. had the following inventories on March 1: Finished Goods ........................... $15,000 Work in Process ........................... 19,070 Materials...
-
In 2012, Forbes magazine listed Bill Gates, the founder of Microsoft, as the richest person in the United States. His personal wealth was estimated to be $66 billion. Given that there were about 309...
-
There are many stereo-isomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane. One of those stereo-isomers undergoes E2 elimination thousands of times more slowly than the other stereo-isomers. Identify...
-
Why does a technical communicator need to know all of this? Isn't there project management software that handles it? Find three online project management sites and print the home page of each. Which...
-
Compare and Contrast the plot of a Rose for Emily vs Plot the Flowers by Alice Walker.
-
A company disburses a total of Tsh 50,000,000 per year on cash. It costs Tsh 75 on average every time securities are sold for cash. The treasury manager estimates that the variance of change in daily...
-
Body-Solid Inc. manufactures elliptical exercise machines and treadmills. The products are prouced in its Fabrication and Assembly production departments. In addition to production activities,...
-
In March 2023, Kuni asks you to prepare his Federal income tax returns for tax years 2020, 2021, and 2022. In discussing this matter with him, you discover that he also has not filed for tax year...
-
1. Describe the impact international trade has on the U.S. economy? weak or stable? 2. Create a list of the different sources of electricity and their prices. 3. Describe the types of energy...
-
Angel has seven DVD movies on a shelf: 4 dramas, 2 science fiction movies, and 1 comedy. Two movies will be selected at random. Determine the probability of selecting each of the following (a) With...
-
Graph one period of each function. y = 4 cos x
-
How many absorptions would you expect S)-malate, an intermediate in carbohydrate metabolism, to have in its NMR spectrum?Explain. (S)-Malate
-
Each of the following compounds has a single 1H NMR peak. Approximately where would you expect each compound toabsorb? (c) (b) (a) "H (d) CH2CI2 (e) O ( N-CH C-C
-
Identify the different kinds of non-equivalent protons in the following molecule, and tell where you would expect each toabsorb. CH-CH C
-
Tim Drinking, Inc. used a predetermined overhead allocation rate to allocate of indirect costs to the Filling Department and the Packaging Department journal entry to record the allocation of...
-
What are the key principles of management, and how do they contribute to effective organizational practices?
-
Discuss the impact of globalization on small businesses in developing countries. Analyze the advantages and disadvantages of globalization for small businesses, and provide recommendations for...

Study smarter with the SolutionInn App


