Propose an efficient synthesis for each of the following transformations: (a) (b) (c) (d) (e) (f) (g)
Question:
(a)
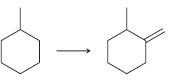
(b)
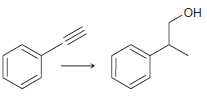
(c)
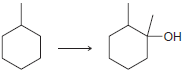
(d)
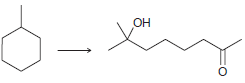
(e)
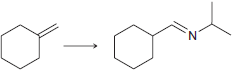
(f)
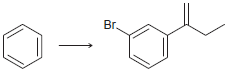
(g)
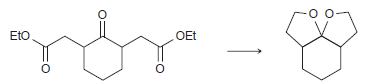
Transcribed Image Text:
-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
a b c d e f g 1 ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose an efficient synthesis for each of the following compounds using the acetoacetic ester synthesis. (a) (b) (c) (d)
-
Propose an efficient synthesis for each of the following transformations: a. b. c. d. Br
-
Propose an efficient synthesis for each of the following transformations. Show all intermediate products, reagents and conditions. NO2 NH2 OH .CN
-
In Problems, use a calculator to give a decimal approximation of the numbers to three decimal places. e 4
-
On September 30, 2016 Sternberg Company sold office equipment for $12,000. The equipment was purchased on March 31, 2013 for $24,000. The asset was being depreciated over a five-year life using the...
-
Describe the theory of organizing.
-
Consider a payoff \(C\) that will occur in 2 years, taking one of the three possible values \(C_{0}, C_{1}, C_{2}\). The short rate lattice for these 2 years is shown in Figure 16.16, with \(d_{i...
-
Consider a blending tank that has the same dimensions and nominal flow rates as the storage tank in Exercise 2.13 but incorporates a valve on the outflow line that is used to establish flow rate w3....
-
In what ways do decentralized organizational structures, characterized by autonomous decision-making units and distributed authority, foster agility and innovation in response to rapidly evolving...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Predict the major product of each reaction below: (a) (b) (c) RCO3H RCO, ? H RCO3H
-
Using any compounds of your choosing, identify a method for preparing each of the following compounds. Your only limitation is that the compounds you use can have no more than two carbon atoms. For...
-
(a) When the switch is closed in the circuit shown in Figure 21-57, does the total power dissipated in the circuit increase, decrease, or stay the same? Figure 21-57, (b) Choose the best explanation...
-
Consider two electrons. They repel. This repulsive force in magnitude is 4.00 N Find distance between the electrons Fibd distance when force is 1.00N
-
You are hiking to the top of Mt. Mitchell, NC (elevation 2 km). You start the hike at 0.5 km elevation where the air temperature is 20 C. There are cumulus clouds overhead with the cloud base at 1.5...
-
An uncharged 6.6-F capacitor is in series, through a switch, with a 4.10-MQ resistor and a 11.6-V battery (with negligible internal resistance.) The switch is closed at t = 0 and a current of /;...
-
1. A point on the third nodal line from the center of an interference pattern is 35 cm from one source and 42 cm from the second source. The sources are 11.2 cm apart and vibrate in phase at 10.5 Hz....
-
1. A magnetic field of 0.0200 T [up] is created in a region. (5 marks) a) Determine the magnitude and direction of the initial magnetic force on an electron initially moving at 5.00 x 106 m/s [N] in...
-
Refer to the following table, which shows the type and manufacturer of vehicles at a specific car and truck dealership. If one person selects a vehicle at random from the dealership, and if each...
-
Selected condensed data taken from a recent statement of financial position of Morino Ltd. are as follows. MORINO LTD. Statement of Financial Position (partial) Other current assets...
-
Zingiberene, a fragrant compound isolated from ginger, has the molecular formula C15H24 and is known not to contain any triple bonds. (a) What is the index of hydrogen deficiency of zingiberene? (b)...
-
Carbonyl groups also count for a unit of hydrogen deficiency. What are the indices of hydrogen deficiency for the reactant and for the product in the equation shown at the beginning of Section 4.16...
-
Write a bond-line formula for each of the following compounds: (a) 1, 4-Dichloropentane (b) sec-Butyl bromide (c) 4-Isopropylheptane (d) 2, 2, 3-Trimethylpentane (e) 3-Ethyl-2-methylhexane (f) 1,...
-
The highest WACC is 11%, the baseline WACC is 7.4% and lowest WACCS is 2.8%. To derive a WACC that better accounts for extreme scenarios, you decide to assign the following probability weights: High-...
-
Give 2 examples of the ff: Statutory law Regulatory law Common law Include the title of each example and explain why you chose these examples ( 1 to 2 sentences ) . Note: Write your reference ( s )
-
The Adams family includes a financially well-informed couple, both aged 36, and two children aged 4 and 6. The family is financially sound but suffered badly during the tech meltdown in 2000. The...

Study smarter with the SolutionInn App


