Propose an efficient synthesis for each of the following compounds using the acetoacetic ester synthesis. (a) (b)
Question:
(a)
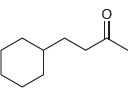
(b)
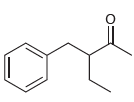
(c)
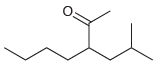
(d)
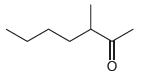
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a b c d ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH H.
-
Propose an efficient synthesis for each of the following transformations: a. b. c. d. Br
-
Describe two methods for determining the molecular mass of a polypeptide. Which is more accurate and why?
-
An independent polling organization interviewed 100 college students to determine their political party affiliations and whether they favor a balanced-budget amendment to the Constitution. The table...
-
The amount of the premium tax credit is tiered based on income and is set so that individuals will not have to pay more than a certain percentage of their income for premiums (see Table 11.1). The...
-
We generated Figure 2.16 by first computing the mean values for each column and then pivoting the values into a single column of mean values. Replicate the data generation steps and use the...
-
You need to calculate the Average Days on Lot based on MSRP and/or Types using the Inventory data. n. Enter functions into the Average Days on Lot chart area that calculate the following: Insert a...
-
What are the types of non-equity entry modes?
-
using the ABC system how much total manufacturing overhead cost would be assigned tp product Y? Using the ABC system how much total manufacturing overhead cost would be assigned to product Z? using...
-
In Problem 22.38, we saw an intramolecular example of a malonic ester synthesis using excess base and 1,4-dibromobutane. If this dibromide is used in an acetoacetic ester synthesis, an intramolecular...
-
Guanidine is a neutral compound but is an extremely powerful base. In fact, it is almost as strong a base as a hydroxide ion. Identify which nitrogen atom in guanidine is so basic, and explain why...
-
Construct the pseudoinverse of A. Begin by using a matrix program to produce the SVD of A, or, if that is not available, begin with an orthogonal diagonalization of A T A. Use the, pseudoinverse to...
-
The Light Record Corporation has been on the calendar year since its inception five years ago. It wishes to change to an April 30 natural business year. For 2019, the calendar year of proposed...
-
Assume the same facts as the problem above except the premium is for coverage from August 1, 2019 to July 31, 2021. What is the largest deduction Wagner Co. can take in 2019? Data from in problem...
-
Lyle Inc. is a cash-basis, calendar-year taxpayer. In 2019, it changes to the accrual method of accounting, Its 2019 income computed under the accrual method is $75,000. The following information...
-
Do either of the transactions below qualify for installment reporting? If not, why not? a. Credit sales of dealer inventory b. Credit sales of property not held as inventory
-
As part of a study of young, UK-based black and minority business graduates career identities and aspirations, research participants were asked to talk about their future work-family balance. The...
-
Convert the following unsigned binary numbers into their decimal equivalents: (a) 01110101 2 (b) 1101.101 2 (c) 1000.111 2
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (b) 3-isopropylhexane (c)...
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
In each pair of compounds, which compound has the higher boiling point? Explain your reasoning. (a) octane or 2,2,3-trimethylpentane (b) Nonane or 2-methylheptane (c) 2, 2, 5-trimethylhexane or nonane
-
A bank can either invest money for three months at 4.00% or for nine months at 4.50%. Ignoring actual/360 day count adjustments for the purpose of this question, the three against nine FRA quote the...
-
Shuggy Otis, an executive at Slapfish Corp. (SC) intends to retire in 11 years. SC just announced that it will start depositing $500.00 at the end of each quarter into each of its workers' retirement...
-
On January 1, the Hanover Beverage Company replaced the palletizing machine on one of its juice lines. The cost of the machine was $195,000. The machine's expected life is five years or 480,000...

Study smarter with the SolutionInn App


