Show how you would use a Grignard reaction to prepare each compound below. a. b. c. d.
Question:
Show how you would use a Grignard reaction to prepare each compound below.
a.
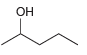
b.
![]()
c.
![]()
d.
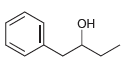
e.
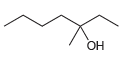
f.
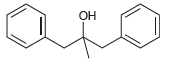
Transcribed Image Text:
OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
a b c d e ...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use a Grignard reaction to prepare the following alcohols: (a) 2-Methyl-2-propanol (b) 1-Methylcyclohexanol (c) 3-Methyl-3-pentanol (d) 2-Phenyl-2-butanol (e) Benzyl alcohol (f) 4-Methyl-1-pentanol
-
Show how you would use a Suzuki reaction to synthesize Bombykol, the sex hormone of the silk moth, from cis-1- bromopent-1-ene and the acetylenic alcohol shown below. Bombykol
-
Show how you would use a Suzuki reaction to synthesize the following biaryl compound. As starting materials you may use the two indicated compounds, plus any additional reagents you need. Make OCH3...
-
Harley-Davidson: Preparing for the Next Century There are very few products that are so exciting that people will tattoo your logo on their body. Richard Teerlink, Retired CEO, Harley-Davidson In...
-
Why are stock market analysts so concerned about what's going on at the Federal Reserve?
-
Using the information from Question 30, what is Oscars basis on his new building? a. $130,000 b. $132,000 c. $140,000 d. $142,000 Question 30 Oscar owns a building that is destroyed in a hurricane....
-
9. Why do executives spend so much time and effort on communicating with noise traders if intrinsic investors ultimately drive a companys share price?
-
Equity method entries. Hanna Company purchased 100% of the common stock of Denver Company on January 2 for $550,000. The common stock of Denver Company at this date was $200,000, and the retained...
-
Morganton Company makes one product and it provided the following information to help prepare the master budget for its first four months of operations: 8 (a) The budgeted selling price per unit is...
-
Miller Toy Company manufactures a plastic swimming pool at its Westwood Plant. The plant has been experiencing problems as shown by its June contribution format income statement below: Contains...
-
Draw a bond-line structure for each of the following compounds: a. 3-Isopropyl-2, 4-dimethyl-2-pentene b. 4-Ethyl-2-methyl-2-hexene c. 1, 2-Dimethylcyclobutene (The name of a cycloalkene will not...
-
In the mass spectrum of bromobenzene (Figure 15.27), the base peak appears at m/z = 77. Figure 15.27 a) Does this fragment contain Br? Explain your reasoning. b) Draw the cationic fragment that...
-
In an attempt to determine whether a relationship exists between the price of a home and the number of days it takes to sell the home, a real estate agent collects data on the recent sales of eight...
-
Based on a survey, assume that 42% of consumers are comfortable having drones deliver their purchases. Suppose that we want to find the probability that when six consumers are randomly selected,...
-
What is the social location that determines this speech community? Is it determined by race, class, gender, sexuality, or some other social location? What makes this speech community unique? What are...
-
Write a program named SumOfNumberOfSquares.java that prompts user to enter a number of integers and calculates the sum of their squares. The following is a sample run. The green fonts represent user...
-
6.4 Charles Augustin de Coulomb was a French physicist who is best known for formulating the law that calculates the force between two electric charges. To honor Coulomb, the unit of electric charge...
-
What amount of cash payments to suppliers will be reported by Indigo Company for the year ended December 31, 2024?
-
Use the fact that a x = a y implies x = y, to solve each equation. 25x = 1252-x
-
What are technical skills At what level are they most important and why?
-
Make a graph of potential energy versus angle of bond rotation for propane, and assign values to the energy maxima.
-
Consider 2-methylpropane (isobutene). Sighting along the C2-C1 bond: (a) Draw a Newman projection of the most stable conformation. (b) Draw a Newman projection of the least stable conformation. (c)...
-
Sight along the C2-C3 bond of 2, 3-dimethylbutane, and draw a Newman projection of the most stable conformation.
-
Justice Corporation Comparative Balance Sheet December 31, 2025 and 2024 2025 2024 Assets Current Assets: $ Cash and Cash Equivalents 2,254 $ 1,876 Justice Corporation reported the following...
-
The Fields Company has two manufacturing departments forming and painting. The company uses the FIFO method of process costing at the beginning of the month the forming department has 33.000 units in...
-
A comparative balance sheet for Lomax Company containing data for the last two years is as follows: Lomax Company Comparative Balance Sheet This Year Last Year $ 96,000 $ 70,000 640,000 672,500...

Study smarter with the SolutionInn App


