In the mass spectrum of bromobenzene (Figure 15.27), the base peak appears at m/z = 77. Figure
Question:
Figure 15.27
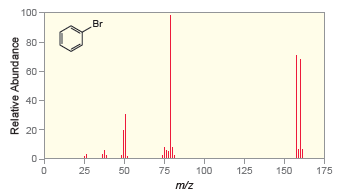
a) Does this fragment contain Br? Explain your reasoning.
b) Draw the cationic fragment that represents the base peak.
Transcribed Image Text:
100 Br 80 60 40- 20 50 25 75 100 125 150 175 m/z Relative Abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
a This fragment is ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
-
The mass spectrum of 1-butanol shows an intense daughter ion peak at m/z 5 31. Explain how this peak might arise.
-
Identify the expected base peak in the mass spectrum of 2, 2, 3-trimethylbutane. Draw the fragment associated with this peak, and explain why the base peak results from this fragment.
-
As speed increases for an object in free fall, does acceleration increase also?
-
What is the role of specialists and traders on the New York Stock Exchange?
-
If Karp had used FIFO instead of LIFO, which of the following ratios computed as of 31 December 2018 would most likely have been lower? A . Cash ratio. B . Current ratio. C . Gross profit margin....
-
Throughout the US presidential election of 2016, polls gave regular updates on the sample proportion supporting each candidate and the margin of error for the estimates. This attempt to predict the...
-
Account balances taken from the ledger of High Flying Logistics Co. on December 31, 2011, follow: Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
3 (a) You are the author of a new novel. You can choose to self-publish by making your own website to with the help of a local developer for a cost of $15,000, or you can sell them via an existing...
-
a. What should the weekly product mix consist of, and what is the weekly net profit? b. Is the optimal solution to question 1 degenerate? Explain your response. c. Is the optimal solution from...
-
Show how you would use a Grignard reaction to prepare each compound below. a. b. c. d. e. f. OH
-
Suppose that the pen-making industry is perfectly competitive. Also suppose that each current firm and any potential firms that might enter the industry all have identical cost curves, with minimum...
-
A car is driving on a straight, level road at constant speed. Draw an FBD for the car, showing the significant forces that act on it.
-
Industrial processes damage the environment and cause serious local and global deterioration. The response in the United States and in most of the developed world has been to adopt a series of fairly...
-
Moxx of Balhoon Ltd is a small car repair company. Its directors are Cassandra and Tobias. On 10th October 2020 it goes into liquidation. Rose Tyler is appointed liquidator. You act for Rose and have...
-
Jonathan, an elderly retired accountant, has amassed a large fortune over the course of his life, as a result of his distinguished career and well chosen investments. In 2017 at the age of 70, he...
-
Lone Oak Brewpub is a brew pub in Charlottetown, Prince Edward Island. They have several taps featuring local beers. They also feature several beers in bottles. Alan Doyle is in Prince Edward Island...
-
(A worker's union from Caesars Entertainment in Windsor, Ontario, which is known as Unifor attempted to reach a deal on their most recent collective agreement on the issues regarding wages,...
-
The ages of the 44 presidents at their first inauguration (as of 2015) are as follows. (Grover Cleveland was elected as both the 22nd and the 24th president. His age at both of his inaugurations is...
-
Listed below are common types of current liabilities, contingencies, and commitments: a. Accounts payable b. Bank loans and commercial paper c. Notes payable d. Dividends payable e. Sales and excise...
-
Draw a Newman projection along the C2-C3 bond of the following conformation of 2, 3-dimethylbutane, and calculate total strainenergy:
-
Identify the functional groups in the following substances, and convert each drawing into a molecular formula (red = O, blue =N): (a) (b) Phenylalanine Lidocaine
-
Give IUPAC names for the following alkanes, and convert each drawing into a skeletal structure: (a) (b) (c) (d)
-
United States Dominance in Psychology Collaboration is a critical feature of global psychology. In order for professionals to work together throughout the world, it is important for them to have some...
-
( support your answer with one academic reference and non-academic or industry reference ). Every year, as a CEO of a company, you are required to make an annual formal report. In your opinion, what...
-
Question 1 Apple is back under the spotlight over labor conditions in its supply chain following an explosive report from The Information on Thursday that revealed new details about the company's...

Study smarter with the SolutionInn App


